Site Monitoring in Clinical Research: What to Expect
Clinical research is a complex, rigorous process designed to ensure the safety and efficacy of new medical treatments, drugs, and devices. A critical component of this process is site monitoring – the systematic oversight of clinical trial sites to ensure that studies are conducted in compliance with the trial protocol, regulatory standards, and ethical guidelines. In this blog, we’ll provide you with an in-depth understanding of site monitoring in clinical research, what to expect, and why it’s essential for successful trials.
You can also learn about remote site monitoring in clinical trials to understand how technology is transforming the process of monitoring from a distance.
What is Site Monitoring in Clinical Research?
Site monitoring is the process by which clinical trial monitors (also known as Clinical Research Associates, or CRAs) visit and oversee clinical trial sites to ensure that the study is being conducted according to the protocol, Good Clinical Practice (GCP) standards, and regulatory requirements. Monitoring is vital for ensuring that data collected is accurate, that patients’ safety is maintained, and that the trial progresses as planned.
Key Objectives of Site Monitoring
Compliance with Protocol: Ensuring that the site follows the study protocol and adheres to ethical standards.
Safety of Participants: Safeguarding the health and safety of clinical trial participants by checking for adverse events and proper documentation.
Data Integrity: Ensuring that all data collected is accurate, verifiable, and reliable.
Regulatory Adherence: Ensuring compliance with local and international regulatory requirements.
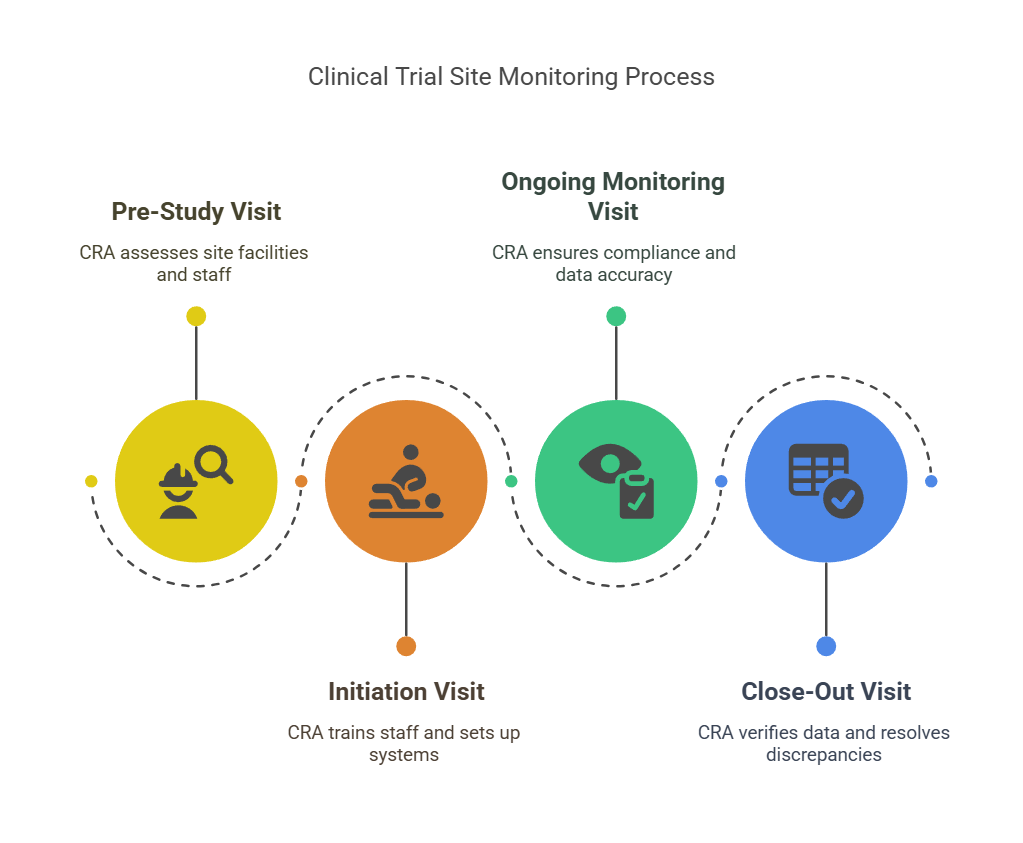
The Site Monitoring Process
The site monitoring process typically involves multiple visits throughout the life of the clinical trial. These visits are conducted by qualified CRAs, who follow a structured process to monitor trial activities. Here's a step-by-step breakdown of the process:
1. Pre-Study Visits
Before the trial begins, CRAs will conduct a pre-study visit (PSV). This visit is essential for evaluating whether the site is equipped to handle the trial. The CRA will:
Assess the site’s facilities and equipment.
Confirm the availability of qualified staff.
Ensure that the site has the proper infrastructure to manage data, patient records, and adverse events.
2. Initiation Visits
Once the study is approved, the CRA will perform the initiation visit to kick-start the clinical trial at the site. The goals of this visit are:
Review the protocol with site staff.
Train investigators and site staff on the study's requirements.
Set up systems for data collection, reporting, and adverse event documentation.
3. Ongoing Monitoring Visits
Throughout the study, CRAs will make regular visits to monitor the progress of the trial. During these visits, they will:
Ensure compliance with the protocol and regulatory requirements.
Review the informed consent process and ensure proper documentation.
Verify patient eligibility and ensure accurate data collection.
Assess the safety of participants by reviewing adverse event reports.
Confirm that the site has adequate record-keeping and reporting procedures.
4. Close-Out Visits
Once the trial is completed, CRAs will conduct a close-out visit to ensure that:
All study data has been collected and properly documented.
All regulatory requirements have been met.
Any remaining queries or discrepancies are resolved.
The site returns all study-related materials, such as drugs, equipment, and documentation.
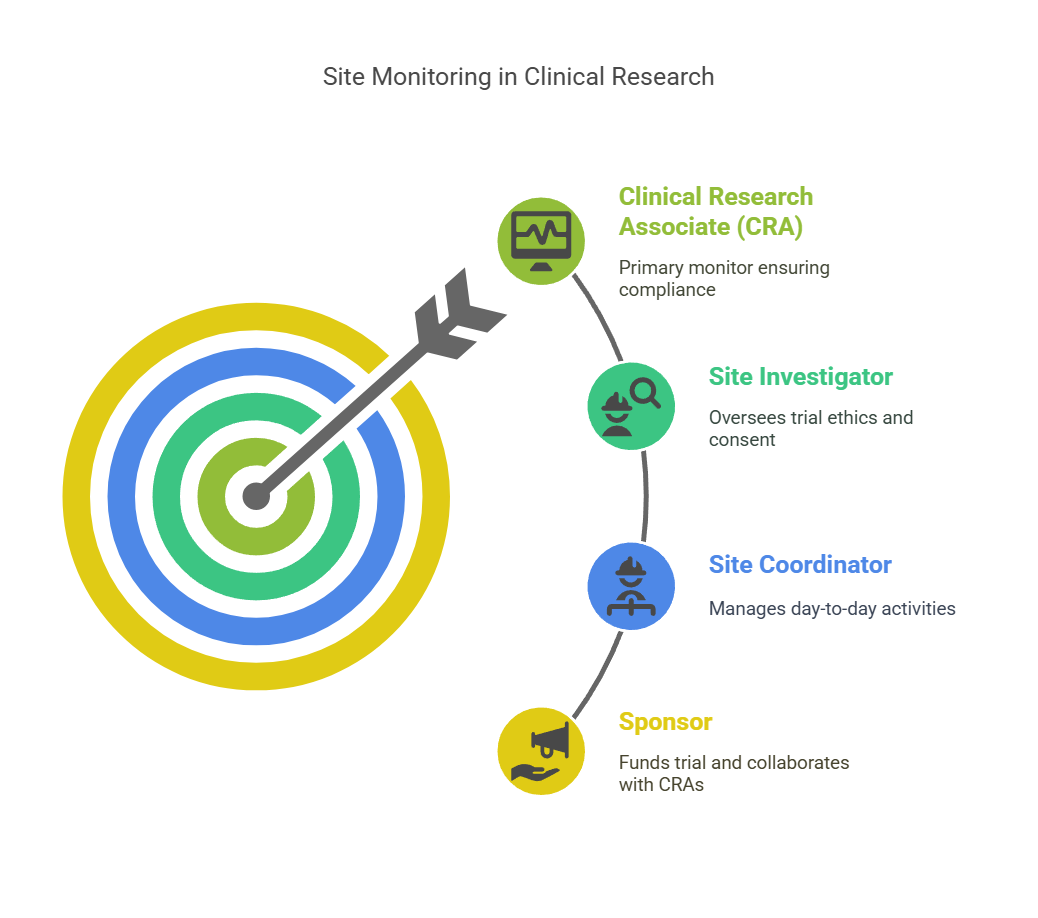
Key Roles Involved in Site Monitoring
Several stakeholders play a role in site monitoring during clinical research. These roles ensure that trials are conducted efficiently, ethically, and according to regulations.
1. Clinical Research Associate (CRA)
The CRA is the primary person responsible for monitoring the site. They are the ones who visit the site regularly, ensure compliance, and report findings to the sponsor.
2. Site Investigator
The site investigator is typically a medical professional responsible for overseeing the clinical trial at the site. They ensure that the study is conducted ethically and are responsible for obtaining informed consent from participants.
3. Site Coordinator
The site coordinator supports the investigator by managing day-to-day activities, such as patient recruitment, scheduling visits, and ensuring proper documentation.
4. Sponsor
The sponsor is the organization (often a pharmaceutical company or research institution) that funds the clinical trial. They work closely with CRAs to ensure that the trial is progressing correctly.
Pro Tip: For a deeper understanding of the role of a clinical site monitor, explore the responsibilities and challenges that come with the position.
What to Expect During a Site Monitoring Visit
A site monitoring visit may seem daunting to clinical trial site staff, but with preparation, it can be a smooth and efficient process. Here’s what you can expect during a typical site monitoring visit:
1. Preparation
Before the visit, the CRA will send out an agenda outlining the key activities they will review, such as patient records, data collection forms, and regulatory documents.
2. Protocol Review
The CRA will review the study protocol with the site staff to ensure that the procedures are being followed correctly. This may involve discussing the objectives, dosing, inclusion/exclusion criteria, and informed consent process.
3. Data Verification
One of the CRA's main tasks is to verify the accuracy of the data collected. They will cross-check data collected from patients with source documents, such as patient charts and lab results. This is to ensure data integrity and identify discrepancies.
4. Patient Safety Review
The CRA will review adverse event reports and ensure that proper action has been taken in response to any incidents. The safety of participants is always the top priority during site monitoring.
5. Regulatory Compliance
The CRA will check that the site maintains proper documentation, including regulatory approvals, ethics committee submissions, and informed consent forms. They will also ensure that the site follows proper protocol deviations and reporting guidelines.
Challenges in Site Monitoring
Despite its importance, site monitoring is not without its challenges. Some of the most common hurdles in site monitoring include:
1. Site Staffing Issues
Shortages in qualified staff can make it difficult to manage the trial and comply with regulatory requirements. Frequent turnover at the site can also lead to delays in study progress.
2. Data Discrepancies
Inconsistent or inaccurate data can arise from errors in patient documentation, data entry, or miscommunication between site staff and the CRA.
3. Site Compliance
Ensuring that a site consistently follows the protocol can be challenging. Non-compliance can be a significant issue, especially if deviations are not reported or handled properly.
Best Practices for Successful Site Monitoring
To ensure successful site monitoring, clinical trial sites, and monitors should adhere to best practices to maintain high standards and compliance. Here are some tips:
1. Clear Communication
Establishing open lines of communication between the CRA, site staff, and the sponsor is essential. Regular meetings, both in-person and virtual, can help address any challenges quickly.
2. Thorough Training
Ensuring that site staff are well-trained on protocol requirements and data collection standards is essential to avoid errors and miscommunication.
3. Regular Audits
Conducting regular internal audits of the site’s processes can help identify and resolve issues before a CRA visit. Audits should include reviewing regulatory documents, patient records, and data accuracy.
4. Proactive Problem-Solving
Addressing issues proactively, such as identifying potential protocol deviations or data discrepancies before a site monitoring visit, can save time and ensure a smoother trial.
10 Less Commonly Known Facts about Site Monitoring
Site Monitoring Reduces Risks: Regular site monitoring helps identify risks early and implement corrective actions, preventing potential protocol violations and enhancing patient safety.
Regulatory Inspections: A site’s performance in clinical trials can impact its ability to conduct future research, as regulatory bodies often use site monitoring results during inspections.
Remote Monitoring: With advancements in technology, some aspects of site monitoring, such as data verification and document review, are increasingly being done remotely, saving time and travel costs.
Cost of Monitoring: Site monitoring is one of the largest costs of conducting a clinical trial. However, investing in thorough monitoring helps ensure compliance and reduces risks.
Monitoring Frequency Varies: The frequency of site monitoring visits can vary depending on the trial phase, risk profile, and sponsor's requirements.
Risk-Based Monitoring: A risk-based approach to monitoring focuses resources on sites or participants with higher risk, allowing for more efficient use of time and budget.
Training is Key: A well-trained study team and site staff significantly reduce the chances of monitoring visits revealing protocol violations or data discrepancies.
Monitoring Systems: Some trials use sophisticated software to track data and site performance, streamlining the monitoring process and ensuring compliance.
Centralized vs. Decentralized Trials: In decentralized trials, monitoring might take place remotely, reducing site visits but requiring advanced tools to manage data from multiple locations.
Site Monitoring and Patient Recruitment: Effective site monitoring ensures that patient recruitment procedures are efficient and meet the trial's eligibility criteria.
Conclusion
Site monitoring is a crucial component of clinical research that ensures the integrity of the trial and the safety of participants. By understanding the monitoring process, the roles involved, and what to expect during visits, sites can ensure they remain compliant and successful throughout the duration of the clinical trial.
At CCRPS, we offer extensive clinical research training to help you master the skills needed for effective site monitoring and other key aspects of clinical trials. Whether you're a beginner or an experienced professional, our clinical research certifications are designed to give you the expertise needed to excel in this field.
-
The CRA is responsible for overseeing trial sites, ensuring protocol compliance, verifying data integrity, and ensuring participant safety during clinical trials.
-
The frequency of site visits depends on the trial phase, risk, and sponsor requirements. They typically occur at regular intervals to ensure compliance.
-
The CRA reviews regulatory documents, informed consent forms, patient records, and data collection sheets to ensure accuracy and protocol adherence.
-
Site monitoring ensures that adverse events are reported, safety protocols are followed, and participants receive appropriate care throughout the study.
-
Non-compliance can result in corrective actions, retraining of site staff, or, in severe cases, the suspension of the trial at that site.
-
A close-out visit marks the end of a clinical trial at a site. The CRA ensures that all data has been collected and regulatory requirements have been met.
-
Yes, with the help of technology, aspects like data verification and documentation reviews can be conducted remotely, improving efficiency.
-
Site monitoring ensures compliance, protects participant safety, maintains data integrity, and meets regulatory requirements, all of which are crucial for trial success.