Scientific Communication & Presentations: MSL Best Practices
Scientific communication is where strong MSLs turn “nice meeting” into measurable scientific impact. When you can translate complex evidence into clear, credible narratives—without overselling, oversimplifying, or drifting off-label—you become the person KOLs trust, medical leaders rely on, and cross-functional teams follow. This guide is built to sharpen the MSL presentation craft that actually moves science forward: rigorous message architecture, evidence handling, question control, objection navigation, and compliant communication under pressure—so your talks sound like science, not marketing.
1) The MSL presentation job is not speaking — it’s scientific risk control
MSLs don’t “present slides.” You manage scientific meaning in high-stakes rooms. Every discussion you lead can either increase trust (and scientific adoption) or create doubt (and compliance risk). The best MSLs are trusted because they consistently deliver four things:
Scientific fidelity: you represent the evidence accurately, including uncertainty, limitations, and nuance.
Clinical relevance: you translate endpoints, designs, and patient populations into practical implications (without claiming beyond data).
Compliance discipline: you stay on-label and avoid promotional tone, steering questions appropriately.
Dialogue control: you can handle tough Q&A without becoming defensive, vague, or speculative.
If you’ve ever felt the pressure of: “I’m not sure what they’re asking,” “They’re pushing me off-label,” “My deck is too dense,” “I lost the room,” or “That one KOL dominated the conversation”—this article gives you the system to fix it.
Strong scientific communication also depends on foundational trial literacy: endpoints, bias controls, and study governance. Your credibility rises when you can connect discussion points to the logic behind primary vs secondary endpoints, the purpose of randomization techniques, and how blinding protects validity. If a study includes safety oversight, you should speak confidently about structures like a Data Monitoring Committee (DMC) without getting lost in jargon.
| MSL Moment | Best For | What “Excellent” Looks Like | Risks / Failure Modes | Best Practice Play |
|---|---|---|---|---|
| Opening framing | Trust in first 60 seconds | States purpose, audience relevance, and boundaries | Sounds promotional or vague | “Here’s the question, evidence, limits, and how we’ll discuss it” |
| Audience calibration | Mixed expertise rooms | Adapts depth without losing rigor | Too basic or too dense | Ask 2 diagnostic questions before the deep dive |
| Claim discipline | Any efficacy/safety point | Claims match data + population + endpoint | Over-interpretation | Use the “PICO lock”: population, intervention, comparator, outcome |
| Endpoint translation | Clinical meaning | Explains endpoint choice + limits | Cherry-picking endpoints | Anchor to endpoint hierarchy + sensitivity analyses |
| Primary vs secondary hierarchy | Credibility | Clear hierarchy, controls, multiplicity context | Inflating secondary outcomes | Separate “signal” vs “confirmed” outcomes explicitly |
| Randomization explanation | Bias concerns | Explains allocation and why it matters | Hand-waving design | Describe randomization method + stratification rationale |
| Blinding clarification | Validity discussion | Explains who was blinded and why | Confusing or inaccurate | State: participants / investigators / assessors / analysts |
| Comparator positioning | Real-world relevance | Explains comparator choice implications | “Apples to oranges” debates | Map comparator to guideline practice and feasibility |
| Safety narrative | Risk–benefit | Uses rates, exposure, seriousness, timelines | Downplaying safety | Separate common AEs, SAEs, AESIs, discontinuations |
| AE definitions consistency | Q&A precision | Uses standard terms correctly | Mixing AE/SAE/AESI | Define once; keep a “terms card” ready |
| Subgroup handling | Precision questions | Pre-specified vs exploratory made explicit | P-hacking accusations | Label subgroup strength; show interaction p-values if available |
| Statistical humility | Uncertainty | CIs, power, missingness discussed plainly | Overconfidence | Use “what data supports” vs “what remains unknown” |
| Study limitations | Trust building | Names real limitations without apologizing | Hiding weaknesses | “Limits → impact → mitigation → next evidence” |
| Real-world evidence positioning | External validity | Separates RCT vs RWE claims | Conflating evidence types | Explain confounding and why RWE answers different questions |
| Slide density control | Retention | 1 insight per slide; readable plots | Text walls | “Headline + 3 bullets + one figure” rule |
| Data visualization | Comprehension | Explains axes, n, timeframe, censoring | Misread graphs | Walk the figure in 20 seconds: what/why/so-what |
| Objection handling | Pushback moments | Validates concern, returns to evidence | Defensive tone | “Agree–clarify–evidence–boundary–next step” |
| Off-label pivot | Compliance | Respectful boundary + proper route | Accidental promotion | “I can’t discuss that here; I can connect you to med info” |
| Hostile questions | High-stress rooms | Calm, specific, no speculation | Over-talking | Answer in 20–40 seconds; offer follow-up evidence |
| KOL dominance | Group discussions | Balances voices | Others disengage | Invite quieter experts by name with a targeted question |
| Handling uncertainty | Gaps in data | Admits unknowns without losing authority | Bluffing | “What we know / don’t know / what’s being studied” |
| Scientific storytelling | Persuasion without hype | Problem → design → evidence → implications | Marketing tone | Use clinical questions as chapter headings, not slogans |
| Time management | Busy clinicians | Hits the 3 critical points early | Running out of time | “Front-load value” + modular sections you can skip |
| Closing & next steps | Action | Summarizes evidence + offers resources | No outcome | Agree on follow-up: paper, data cut, med info request |
| Post-meeting documentation | Continuity | Captures insights & questions compliantly | Lost intelligence | Structured note: question, evidence referenced, follow-up owner |
| Cross-functional handoff | Impact scaling | Scientific insights routed correctly | Noise to internal teams | Insights tagged: safety, endpoints, access, evidence gaps |
| Conference presentation support | Visibility | Aligns on messaging, not promotion | Brand drift | Ensure claims align with label & published evidence boundaries |
2) Build your scientific message architecture (so you never ramble)
The fastest way to sound “junior” is to present facts without structure. Senior MSL communication is architecture: clear questions, correct evidence boundaries, and a storyline the audience can follow under cognitive load.
Use the “Clinical Question Spine” (CQ Spine)
Every strong MSL presentation can be reduced to five sentences:
What clinical problem are we solving? (context + unmet need)
What evidence question did the study ask? (design intent)
What did the data show? (results with correct boundaries)
What does it mean in practice? (implications, not exaggeration)
What remains unknown? (limitations, next evidence)
When you build your talk around questions rather than “slides,” you don’t get lost. You also become harder to bait into off-label drift because your boundaries are explicit.
To strengthen this spine, anchor key explanations to trial fundamentals: why endpoints matter (primary vs secondary endpoints), how bias is controlled (randomization, blinding), and what oversight protects patients and data integrity (DMC roles).
The “Claim Ladder” (to avoid overreach)
Build your statements on a ladder:
Observed (what happened in the data)
Supported (what the design allows you to infer)
Hypothesized (what might be true, but is not proven)
Unknown (what you must not imply)
Most MSL mistakes happen when people jump from observed → generalized practice. Your authority increases when you can say, “This is what the data supports in this population, under this comparator, for this endpoint.”
A practical rule: “If you can’t name the population, don’t make the claim.”
If you can’t quickly restate inclusion/exclusion and context, you risk speaking outside the evidence. Keep a “population card” in your notes: indication, severity, prior lines, comorbidities, baseline risk.
For basic study literacy reinforcement, CCRPS frameworks like clinical trial protocol essentials and broader standards (e.g., ICH guidelines simplified) help you speak with disciplined clarity.
3) Evidence handling under pressure (Q&A, objections, and “push” moments)
Great MSLs don’t just “know the data.” They can handle pressure without losing scientific accuracy.
A. The 20–40 second answer format
Long answers sound uncertain. Tight answers sound expert.
Use:
One sentence: direct answer (what evidence supports)
One sentence: boundary (population/endpoint limitation)
One sentence: implication (what it suggests, not promises)
Offer follow-up: “I can send the paper / connect to med info”
This prevents you from wandering into speculation.
B. Objections: validate, then return to evidence
If someone says “This isn’t clinically meaningful,” don’t fight. Use:
Validate: “That’s a fair concern.”
Clarify: “Do you mean effect size, durability, or applicability?”
Evidence: present the relevant endpoint and context
Boundary: explain what the data can’t claim
Next step: agree what additional evidence they’d want
This approach becomes especially important when discussions drift into safety. A clean vocabulary based on AE definitions and management prevents sloppy language that damages credibility.
C. The “off-label pivot” that still feels helpful
The wrong move is “I can’t talk about that” and stopping. The right move is:
Acknowledge the intent (why they asked)
Set boundary
Offer the compliant route (medical information request, published data, ongoing studies)
Offer to follow up with what you can provide
This preserves trust while protecting compliance.
What’s your biggest MSL presentation blocker right now?
4) Presentation execution: how elite MSLs run the room
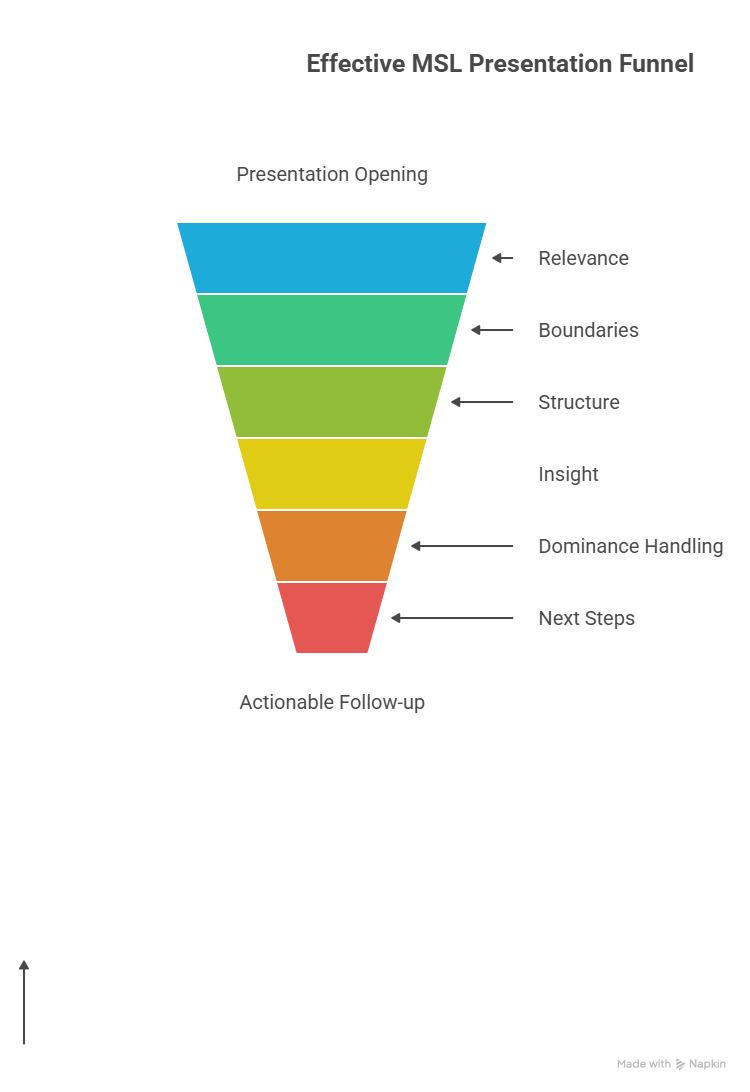
A. The first 3 minutes decide whether the room trusts you
Your opening must do three jobs fast:
Establish relevance (“why this matters clinically”)
Establish boundaries (“what the data can and can’t claim”)
Establish structure (“how we’ll walk through it”)
A clean way to do this is to preview your CQ Spine. It sounds confident because it is.
B. “One insight per slide” is not a style choice — it’s respect for cognition
If you need 10 bullets to make a point, you don’t have a point. You have a data dump.
Make each slide do one job:
define the question
show the design logic
show the result
show the limitation
show the implication
When you show a figure, narrate it in 20 seconds:
what you’re looking at
what changed
why it matters
what it doesn’t prove
This is especially important for endpoints and safety outcomes (again, don’t drift from endpoint hierarchy and use precise AE language aligned with AE identification and management).
C. Handling dominance without disrespect
If one KOL dominates, don’t cut them off. Redirect with precision:
“That’s helpful context. I want to pull in two other perspectives.”
Ask a targeted question to a quieter person: “Dr. X, in your population, what would be clinically meaningful here?”
That’s leadership. It also prevents the meeting from becoming a debate you can’t win.
D. Convert the meeting into a next step (without being salesy)
Your close should not be “Any questions?” Your close should be:
“Here are the 2–3 takeaways the data supports.”
“Here’s the boundary / what we don’t know yet.”
“If it’s useful, I can follow up with: the paper, a figure pack, or a med info request pathway.”
This is how you create follow-up without promotional tone.
5) Post-presentation impact: turn scientific communication into career leverage
You can be excellent in the room and still fail to create impact if you don’t capture and route insights.
A. Capture insights like a scientist, not a diarist
Your post-meeting note should be structured:
Question asked (in neutral language)
Evidence used (publication / dataset reference)
Boundary stated (what was not claimed)
Follow-up requested and owner
Tag the insight (safety, endpoints, access, evidence gap)
This prevents “lost intelligence” and makes you valuable internally.
B. Build a “question bank” and rehearse your top 12
Most MSLs see the same hard questions repeatedly:
subgroup relevance
comparator choice
endpoint meaningfulness
safety tradeoffs
generalizability
missing data / adherence
off-label curiosity disguised as “scientific”
Prepare the best 12 answers with boundaries. You’ll sound senior fast.
C. Use CCRPS resources to strengthen your scientific foundations
If you ever feel shaky on trial fundamentals, revisit:
protocol structure and intent (clinical trial protocol guide)
why bias controls exist (randomization, blinding)
oversight and governance (DMC roles)
safety vocabulary (AE management, drug safety reporting timelines)
These links help keep your scientific communication disciplined and defensible.
6) FAQs: Scientific Communication & Presentations for MSLs
-
Be persuasive through clarity and boundaries, not hype. Use the CQ Spine, state limitations, and separate “observed” from “inferred.” This increases trust.
-
Answer what you can, state what you don’t know, and offer a concrete follow-up route. Avoid speculation. “I’ll confirm and send the source” beats improvisation.
-
Define the endpoint in plain language, state why it matters, then state the limitation. Anchor to primary vs secondary endpoints so hierarchy is clear.
-
Validate the concern, clarify the specific critique, then respond with design logic (randomization, blinding, comparator choice). Keep tone calm and evidence-first.
-
One insight per slide. Use headlines that state the takeaway, not the topic. If a figure is complex, add a 1-sentence interpretation and narrate it in 20 seconds.
-
Summarize the question, evidence, boundary, and next step. Keep it scientific and specific—no marketing tone.