How to Progress Your PV Career By Joining The Right Company
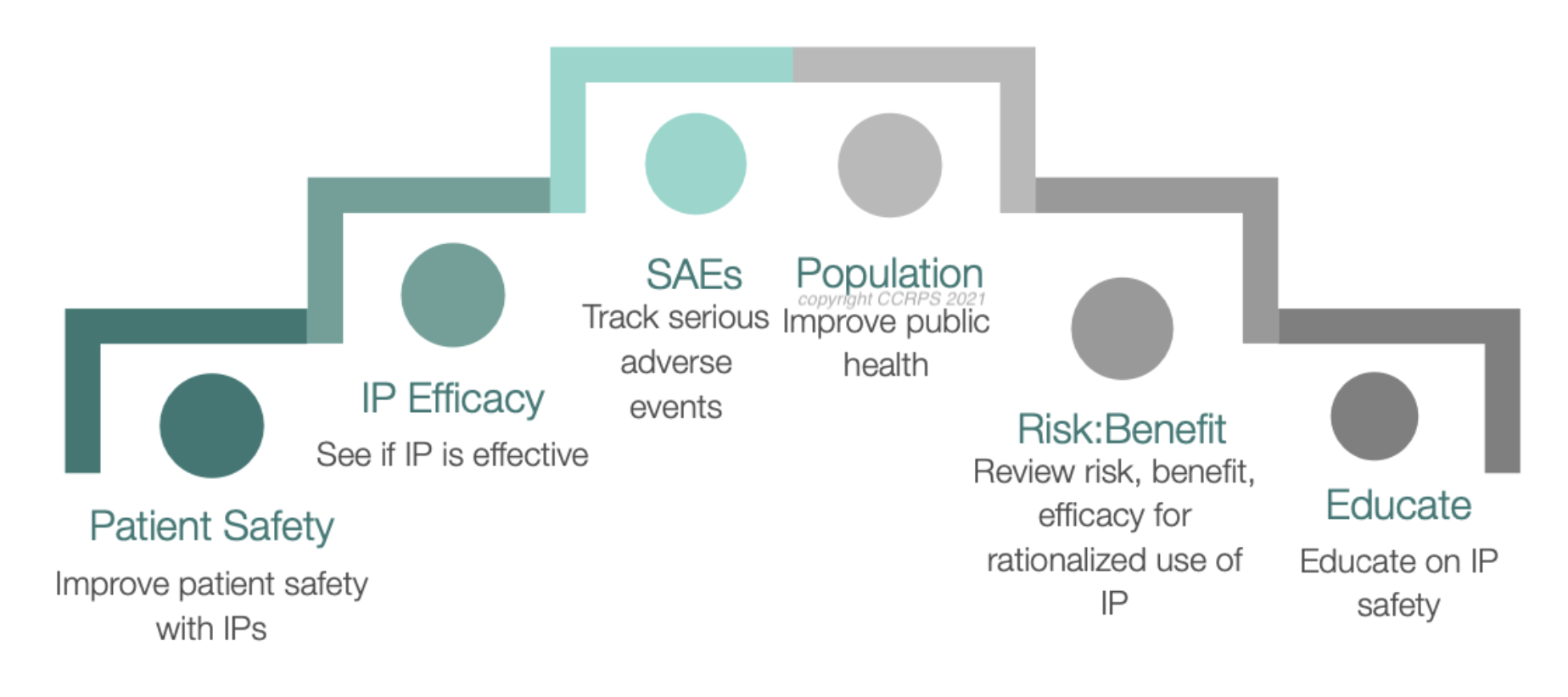
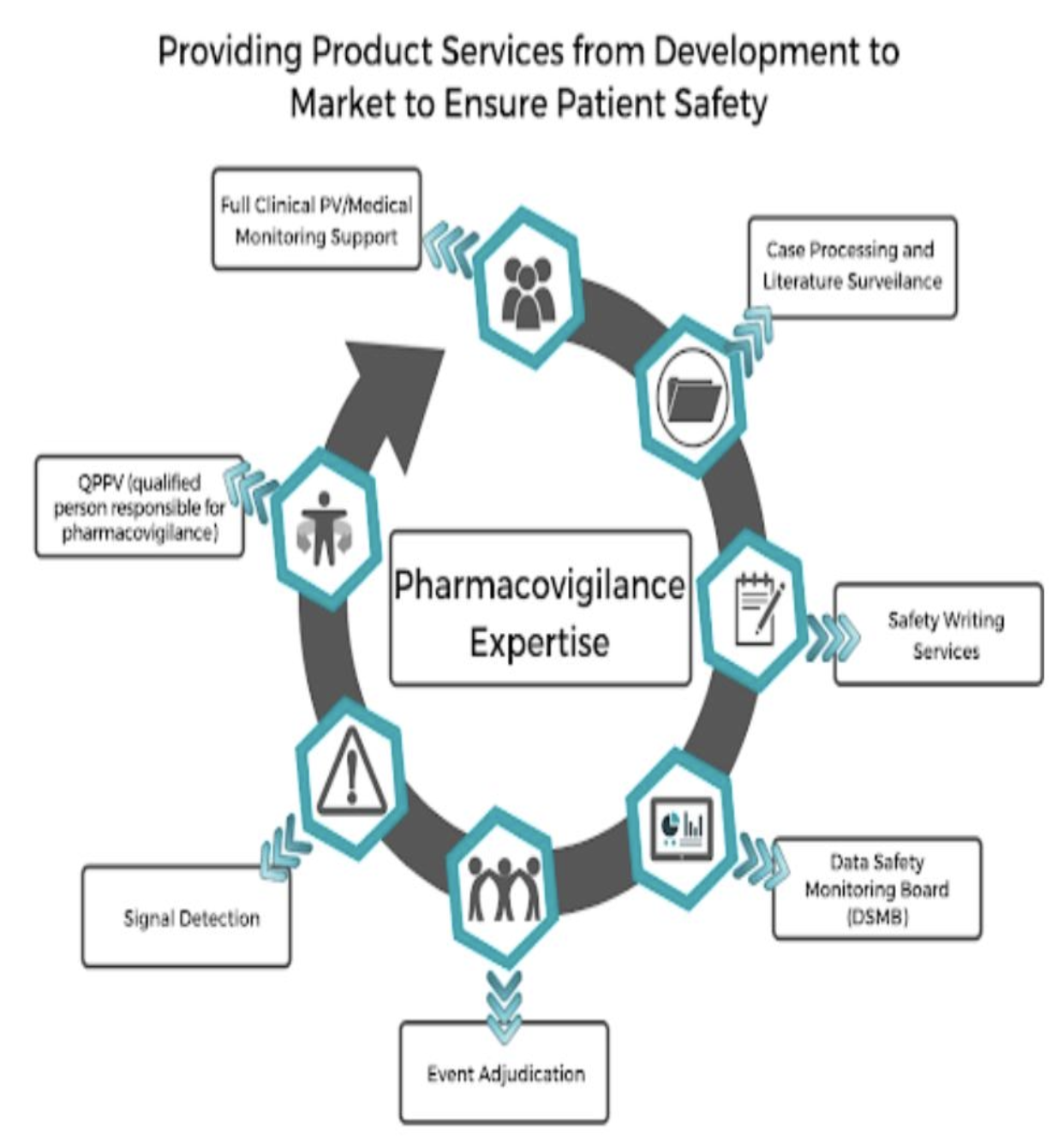
Pharmacovigilance (PV), or drug safety, is the study of a drug’s effects. Most importantly, PV monitors a drug’s adverse effects and protects consumers. PV professionals often find intellectually and financially fulfilling careers in the booming clinical research and pharmaceutical industry. Many start in the field in case management positions, but find it hard to access higher positions such as line management or technical specialists. Senior PV positions can be very competitive and difficult to acquire. However, doing research and planning ahead while you are applying for your first job can help you overcome these obstacles. Here are some tips on how to progress your career in PV.
Join the right company
When companies are hiring for senior positions, candidates must have the right qualifications and experience. However, critical experiences for higher positions can be hard to come by, especially in larger companies where case processing teams rarely work with senior level staff. When PV professionals feel that there aren't enough opportunities for growth in their work environment, one of the most effective strategies is to find and join a different kind of company. While this might seem drastic, making the change early can have lasting benefits on your career.
Here are ways your workplace shape your career:
Joining a smaller company and team will allow you to juggle more roles and gain the experience you need to apply for senior positions.
Joining a company where case processing occurs at the headquarter will allow you to work and network with more senior staff, which can prove extremely advantageous in the long run.
Joining a generics company could be the right move for you, especially if you are interested in senior technical roles. Since a generics company works with compounds that have expired patents and have been on the market for a considerable time, developing generic drugs carries less risk. Therefore, you are more likely to be promoted into signal detection work with less experience.
Joining regulatory or drug safety consultancies like CROs could also be smart for those interested in technical roles. They often offer job training for case processors to transition into signal detection roles. Additionally, since these companies specialize in outsourcing, working there would allow you to gain diverse product experiences and help your resume stand out.
Joining a health authority like the FDA or the EMA would allow you to read and analyze drug evaluations submitted by pharmaceutical companies. Your understanding on how to get approved by the regulatory bodies is a highly desirable skill set for pharmaceutical companies.
Advance Your Career in Pharmacovigilance: Navigating Opportunities in Drug Safety
List of Courses:
While applying for senior PV positions can be competitive and difficult, there are many ways for starting professionals to build their credentials while having an unique and fulfilling career. If you want to learn more about pharmacovigilance, please visit our website at CCRPS.Org. We offer an online pharmacovigilance course to help new professionals improve their qualifications and gain expert insight in the field. The course is curated by real clinical research professionals and perfect for a busy schedule.