Patient Safety Oversight in Clinical Trials: PI’s Essential Role
Patient safety oversight is the PI’s non-negotiable job—even when operations, CROs, and vendors do most of the daily execution. If you don’t design the safety system, ask the right questions, and force clean documentation, you can end up with “everything was done” while the trial still has late SAE reporting, messy narratives, unresolved protocol deviations, and weak escalation records. This guide breaks down exactly how PIs protect participants in real-world trial conditions: the oversight routines, decision triggers, documentation habits, and cross-team governance that prevent safety drift—without turning the PI into an operational bottleneck.
1. The PI’s Safety Oversight Job: What You Must Own vs What You Must Verify
A PI doesn’t “do everything,” but a PI owns the system that prevents safety failures. That means two parallel responsibilities:
Clinical responsibility for participant welfare (medical decisions, risk/benefit judgment, immediate action).
Oversight responsibility for process integrity (making sure safety events are recognized, documented, escalated, reconciled, and reported correctly).
The most common safety failure pattern is not negligence—it’s diffusion of responsibility. The CRC thinks the CRA will catch it, the CRA assumes PV has it, PV assumes the site reported it correctly, and the sponsor assumes “the CRO is on it.” That’s why the PI must enforce clear handoffs, especially when safety intersects with data capture in the CRF workflow and edit checks (CRF best practices) and site documentation habits (CRC responsibilities).
Here’s the clean way to think about “PI oversight”:
You own the safety governance: what gets reviewed, when, how decisions are documented, and who escalates what. That governance must align with monitoring reality (CRA role clarity) and with formal safety functions like pharmacovigilance (PV essentials).
You verify the safety signals: AE/SAE timeliness, severity grading consistency, relationship assessments, protocol deviation patterns, and “quiet signals” that don’t look like emergencies until they cluster.
You control decision rights: dose interruption rules, withdrawal decisions, unblinding pathways (when allowed), and how safety impacts endpoints (primary vs secondary endpoints).
If your trial includes controls, your oversight gets harder: participants and sites behave differently, symptom reporting patterns shift, and safety narratives can get messy when expectations are influenced by assignment. You need the operational awareness of placebo dynamics (placebo-controlled trials) and the process discipline of blinding controls (blinding types & importance) to prevent unintentional bias and unsafe decision shortcuts.
The PI safety mindset: You are not there to “trust the system.” You are there to design it, stress-test it, and audit it in motion—using evidence, not optimism.
2. Safety Oversight Architecture: The Systems a PI Must Set Up Before First Patient In
If safety oversight depends on heroics, it will fail under load. The PI’s smartest move is to build a safety architecture that makes good outcomes the default.
1) Define a safety RACI that names objects, not just people
A weak RACI says “CRC documents AEs, CRA monitors, PV reports.” A strong RACI names the actual objects that move:
AE source note → AE CRF entry → query resolution → PV case creation → reconciliation closure
Deviation detection → deviation classification → safety impact assessment → CAPA → retraining documentation
Consent update → re-consent confirmation → filing → monitoring verification
Your operational partners must understand clinical research roles and their boundaries (CRA responsibilities) and site execution realities (CRC responsibilities). Your safety function must follow PV fundamentals (pharmacovigilance guide), and your data capture must align with robust CRF design (CRF best practices).
PI move: Create a one-page “Safety Handoff Map” that lists every handoff, the owner, the expected time window, and what evidence proves it happened.
2) Pre-build decision triggers (so you don’t improvise under pressure)
When a serious event occurs, teams often waste time debating whether this is urgent. That delay is the safety risk.
Build triggers like:
What constitutes “immediate PI notification” (and what is merely “review next business day”)
When dose interruption is mandatory
When unblinding pathways can be initiated, and who authorizes it (especially critical in blinded designs: blinding overview)
When randomization/IRT issues become safety threats (rule misunderstandings happen—ground your thinking in randomization techniques)
PI move: Turn triggers into a site job aid with three columns: “Trigger → Immediate action → Who to contact.”
3) Align safety oversight to study endpoints and analysis needs
Safety oversight is not separate from “science.” If endpoints are poorly defined, safety decisions get distorted by pressure to “complete assessments” or “hit visit windows.” Tight endpoint understanding reduces unsafe behavior (primary vs secondary endpoints), and a basic stats lens helps you interpret signal noise vs meaningful drift (biostatistics overview).
If your design includes placebo/control conditions, set expectations early about retention, symptom reporting, and ethical guardrails (placebo-controlled trials).
PI move: Write a “Safety vs Endpoint” rule: no endpoint is worth a participant continuing when safety thresholds are crossed. Put that rule in training and meeting notes so it’s provable later.
3. AE/SAE Oversight: How PIs Prevent Late Reporting, Weak Narratives, and PV Breakdowns
Most safety failures show up as documentation failures first: missing onset dates, unclear timelines, “patient felt bad” narratives, and inconsistent causality rationale. Those failures become compliance problems when PV needs structured data for reporting and when auditors ask, “How did you decide this?”
A PI’s oversight should focus on five high-leverage controls:
1) Narrative quality control (QC) using a template
A high-quality safety narrative includes:
timeline (onset, interventions, outcomes)
objective findings (vitals, labs, imaging as relevant)
relationship assessment rationale
dechallenge/rechallenge considerations (when applicable)
protocol-required follow-up plan
This is where CRF discipline matters (CRF best practices) and where CRC execution must be consistent (CRC responsibilities). CRAs should verify source-to-CRF consistency (CRA role clarity), and PV should ensure reporting structure is intact (pharmacovigilance essentials).
PI move: Sample 3–5 AEs/SAEs monthly and score them against a narrative checklist. Track score trends.
2) Timeliness as a system, not a reminder email
Late reporting happens due to weekends, staff changes, uncertainty (“is this serious?”), and messy workflow ownership.
Fixes that work:
a named primary and backup reporter
a 24-hour triage rule for potential SAEs
a weekend/holiday escalation plan
a clear “PI review within X hours” commitment
PI move: Keep a “timeliness dashboard” with simple timestamps: event awareness → site report → PV case submission → follow-up closure.
3) Reconciliation: EDC safety data must match PV cases
If the EDC says AE exists but PV doesn’t, you have systemic failure. If PV has a case but EDC lacks detail, you have a source capture problem. Reconciliation is where oversight becomes real.
This requires clean data flows, which are influenced by randomization and blinding mechanics (especially if event management intersects with treatment knowledge): randomization explained and blinding explained.
PI move: Demand a monthly reconciliation report with three buckets: “matched,” “missing in PV,” “missing in EDC,” plus closure deadlines.
4) Don’t let protocol deviations become hidden safety problems
Deviations are often treated as admin noise, but repeated deviations can signal unsafe processes: missed labs, late assessments, missed follow-up, or medication errors. That’s why deviations must be reviewed for safety impact, not just counted.
Use site ops reality (CRC responsibilities) and monitoring reality (CRA roles) to identify root cause and correct it.
PI move: Trigger retraining when a deviation category repeats twice in a month—not when it becomes a chronic pattern.
5) Avoid “endpoint pressure” safety drift
Teams sometimes subtly pressure participants to “finish the visit” or “complete endpoints” even when safety suggests stopping. That pressure can be fueled by unclear endpoint thinking (endpoints clarity) and weak understanding of trial design ethics (placebo-controlled trials).
PI move: Put a one-line safety-first rule into training, meeting minutes, and escalation notes so it’s documented: Safety overrides schedule.
4. Signal Detection & Escalation: How PIs Catch Safety Drift Before It Becomes Harm
Safety oversight isn’t only about reporting events. It’s about detecting patterns early and escalating appropriately—without panic, without delays, and with documentation that stands up later.
The PI’s “early signal” dashboard (simple, powerful, real)
You don’t need a complicated analytics platform to spot drift. You need a disciplined set of indicators:
AE rate per participant-month (overall and by site)
SAE timeliness distribution (not just “% on time,” but how late and why)
Protocol deviation clusters (category + site + recurrence)
Query patterns in safety fields (suggesting confusion or under-capture)
Lab abnormal trends (outliers and repeated borderline signals)
Because data capture quality drives signal detection, you need CRF alignment (CRF best practices) and basic analytic literacy (biostatistics overview). Because sites behave differently under different designs, keep design ethics in mind (placebo-controlled trials) and protect blinding integrity (blinding types).
PI move: Ask for the dashboard every month—and document your review with two sentences: what you saw and what you did.
When a DMC exists, the PI still has responsibilities
A Data Monitoring Committee is not a substitute for site-level safety oversight; it’s an independent governance layer. PIs should understand:
what data goes to the DMC
how frequently it is reviewed
how decisions are documented and communicated
how local site actions align with global oversight
If your trial uses a DMC, study the structure here: DMC roles explained. If DMC decisions affect study operations, ensure CRAs and CRCs align quickly (CRA roles, CRC responsibilities).
PI move: Maintain a “Safety Decision Log” that captures DMC-related communications, site-level actions, and rationale.
Escalation that works: clarity beats urgency theater
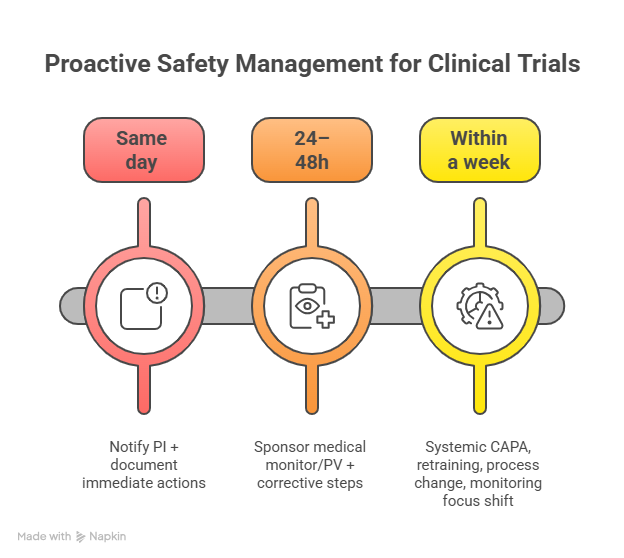
Escalation fails when it’s emotional, vague, or undocumented. A PI should enforce an escalation ladder:
Tier 1 (Same day): notify PI + document immediate actions
Tier 2 (24–48h): sponsor medical monitor/PV + corrective steps
Tier 3 (within a week): systemic CAPA, retraining, process change, monitoring focus shift
This ladder must tie into PV structure (pharmacovigilance essentials) and monitoring accountability (CRA responsibilities).
PI move: Every escalation email should include: impact, evidence, decision needed, deadline, and what happens next if missed.
5. Documentation & Inspection Readiness: The Evidence That Proves You Protected Participants
In inspections and audits, the question isn’t “Did you care about safety?” It’s “Show me the evidence that you controlled safety risks.”
That evidence is built through consistent documentation:
1) Safety oversight notes (short, consistent, defensible)
You don’t need long essays. You need consistent proof:
reviewed AE/SAE listings
reviewed timeliness
reviewed deviations with safety impact
issued retraining/CAPA decisions
confirmed reconciliation outcomes
Tie these artifacts into regulatory document control and filing discipline (managing regulatory documents) and ensure the documentation links back to the CRF/source truth (CRF best practices).
PI move: Create a monthly “PI Safety Review Note” template (5 bullet points). File it consistently.
2) Version control of consent and safety communications
When risk information changes, the consent process must keep up. If versions drift, you have a safety compliance issue.
Support this with:
consent version logs
site acknowledgment of safety letters
retraining evidence when new risk is communicated
This is operationally intertwined with site roles (CRC responsibilities) and monitoring checks (CRA roles).
PI move: Require “acknowledged by site” as a hard requirement for major safety updates.
3) Protect the integrity of blinding and randomization records
In blinded trials, safety decisions can push teams toward informal unblinding. That’s a risk to validity and can even influence care decisions improperly.
Keep your process disciplined by understanding:
blinding risk points (blinding guide)
randomization mechanics (randomization explained)
PI move: Maintain a formal unblinding log with rationale, authorizer, timestamps, and downstream actions.
4) Link safety actions to study endpoints and analysis plans
If a safety decision affects endpoint data (missed assessments, early discontinuation), document how you handled it. This avoids later confusion and credibility gaps.
Use endpoint clarity (primary vs secondary endpoints) and analysis literacy (biostatistics overview) to keep records consistent.
PI move: Document “why” decisions were made, not just “what happened.”
6. FAQs: Patient Safety Oversight in Clinical Trials (PI-Level Answers)
-
To ensure the safety system actually functions: AEs/SAEs are recognized, documented, escalated, reconciled, and acted on—supported by PV structure (pharmacovigilance essentials) and clean CRF capture (CRF best practices).
-
Use a cadence: weekly SAE/deviation review, monthly trend dashboard, and strict triggers for immediate notification. Delegate data gathering to CRCs (CRC responsibilities) and verification to CRAs (CRA roles), but keep decision rights and documentation under PI control.
-
Ambiguous recognition, weekend gaps, unclear ownership, and weak escalation ladders. Fix with a 24-hour triage rule, backup reporters, and time-stamped reporting dashboards aligned with PV expectations (PV guide).
-
Use a structured narrative template that forces timelines, objective data, causality rationale, follow-up plans, and outcomes. Ensure the CRF captures required detail (CRF best practices) and the site team is trained to document consistently (CRC responsibilities).
-
A DMC is independent oversight at the trial level; the PI still runs local safety decisions, documentation, and immediate participant protection. Understand the committee mechanics and decision documentation expectations (DMC roles explained).
-
They shape what information is available and how decisions are made. Poorly controlled unblinding can bias care and data; misunderstood randomization rules can create assignment errors. Keep processes strict and documented (blinding guide, randomization explained).