Risk-Based Monitoring Strategies: CRA Mastery Guide
Risk-based monitoring changed the job of the CRA in a deep way. Monitoring is no longer about treating every site, every subject file, and every data point as if they carry the same threat. Strong monitoring now depends on knowing where quality can actually break, where patient safety can be compromised, where protocol execution can quietly drift, and where site behavior signals trouble before the obvious metrics fully collapse.
That shift rewards judgment. A strong CRA does not just review what happened. A strong CRA learns to identify where risk is building, why it is building, which signals matter most, and what level of intervention will protect study quality without wasting attention on low-value noise. That is the real foundation of risk-based monitoring mastery.
1. Why Risk-Based Monitoring Matters and What It Changed for CRAs
Traditional monitoring logic leaned heavily on uniformity. Visit the site, review the data, check the documents, confirm compliance, and repeat across all sites in similar ways. That model can still create discipline, but it becomes inefficient when studies are larger, data flows are faster, site variability is wider, remote systems are deeper, and risk signals are available much earlier than they used to be. A one-size-fits-all monitoring pattern often wastes time on lower-value checks while higher-risk processes quietly deteriorate in the background.
Risk-based monitoring pushes the CRA to focus on what matters most first. That means patient safety, data integrity tied to critical endpoints, informed consent quality, investigational product handling, protocol compliance in high-impact procedures, adverse event reporting, and the site behaviors most likely to damage study validity. A CRA who still treats all findings as equal will struggle in modern studies because modern studies require prioritization under pressure.
This is why true RBM skill sits on top of stronger foundations in clinical research associate CRA roles, skills & career path, clinical research associate CRA essential monitoring techniques, GCP compliance essentials for clinical research associates, site selection & qualification visits essential guide for CRAs, and investigator site management mastery proven CRA strategies. RBM does not replace core CRA discipline. It sharpens where and how that discipline is applied.
The change also affects how CRAs think about site oversight. A site may look stable because it answers emails, closes queries, and hosts visits politely. Yet the real risk may be building in delayed source entry, repeated near-miss deviations, inconsistent consent sequencing, weak PI oversight, undertrained backup staff, or poor escalation reflexes. Risk-based monitoring trains the CRA to look beyond surface cooperation and ask a harder question: where is the site most likely to fail in a way that meaningfully harms the study?
That matters because study failures rarely begin with one dramatic error. They usually begin with patterns that were visible early but not interpreted well enough. A site misses the same kind of procedural detail across several subjects. A coordinator starts documenting late. AE narratives become thinner. Eligibility review becomes too casual. Source support weakens around the most time-sensitive endpoints. The strongest CRAs learn to see these patterns early and respond before they become findings, major protocol deviations, or credibility problems with the sponsor.
2. How CRAs Should Identify, Prioritize, and Interpret Risk Signals at Sites
Risk-based monitoring begins with the ability to separate true risk from background operational mess. Not every issue deserves the same level of concern. A few administrative delays may be irritating but manageable. A pattern of delayed AE entry, weak eligibility documentation, or inconsistent endpoint execution can threaten the entire reliability of the trial. The CRA’s job is not to react equally to everything. The job is to identify what carries the highest consequence.
The first step is knowing what counts as critical. CRAs should anchor their risk thinking to patient safety, rights, well-being, protocol compliance in study-defining processes, and data integrity tied to critical endpoints. That sounds obvious until a monitor is drowning in site details and starts treating all cleanup work as equal. Strong CRAs keep returning to the highest-value questions. Are subjects being consented properly? Are eligibility decisions defensible? Are safety events captured and escalated correctly? Is investigational product being handled appropriately? Are critical assessments being performed and documented in a way the study can trust?
That thinking becomes much stronger when built on what is good clinical practice GCP a clear practical explanation, informed consent procedures mastering GCP compliance, serious adverse events SAEs definition & reporting procedures, adverse events AEs identification reporting and management, and clinical trial documentation under GCP comprehensive guide. The CRA who understands the study’s critical processes deeply will always interpret site signals better than the CRA who monitors by habit alone.
The second step is reading patterns, not isolated incidents. One missed visit window may reflect a patient problem. Repeated missed windows across multiple subjects may indicate scheduling weakness, poor patient communication, or site overload. One slow query response may be noise. A growing backlog of aging queries may signal weak accountability or failing bandwidth. One consent correction may be salvageable. Several version-control issues point to a regulatory process problem. Risk-based monitoring becomes powerful when the CRA stops asking only “what happened?” and starts asking “what pattern is this pointing to?”
The third step is connecting remote and on-site signals. Modern monitoring often gives the CRA access to query trends, central review findings, data listings, protocol deviation output, remote dashboard alerts, and site communication history well before the next visit. A strong CRA does not wait for the visit to begin thinking. The visit should be shaped by pre-identified concerns. That allows monitoring time to be focused where consequence is highest instead of spread evenly across low-value review.
The fourth step is interpreting behavior around the signal. Sometimes the problem is not the finding itself but how the site responds. A site that acknowledges risk quickly, investigates honestly, and implements specific fixes is very different from a site that minimizes, deflects, delays, or closes issues cosmetically. Risk lives in attitude as much as in errors. A monitor who ignores that will keep getting surprised by recurring failures.
3. Core Risk-Based Monitoring Strategies That Separate Average CRAs From Strong Ones
One of the most important RBM strategies is critical process mapping. Before deep monitoring begins, the CRA should know which protocol procedures, safety processes, data points, and operational workflows are too important to monitor casually. That includes eligibility determination, consent sequencing, primary endpoint assessments, AE and SAE handling, investigational product accountability, protocol-specific timing requirements, and PI oversight in critical decisions. Once that map is clear, the monitor can focus attention where study damage would be highest.
Another defining strategy is targeted source review. Risk-based monitoring does not mean careless monitoring. It means smarter review intensity. The CRA may choose deeper review around critical visits, critical data transitions, safety-relevant subjects, questionable enrollments, or patterns emerging from central data review. This approach becomes more effective when informed by clinical trial protocol the definitive guide with examples, primary vs secondary endpoints clarified with examples, randomization techniques in clinical trials explained clearly, blinding in clinical trials types & importance clearly explained, and data monitoring committee DMC roles in clinical trials explained. The more clearly a CRA understands trial design and endpoint logic, the better the CRA can decide what deserves concentrated attention.
A third strong strategy is site segmentation. Not all sites need the same oversight intensity at the same time. Some sites are stable and responsive. Others are new, understaffed, overconfident, or operating in a way that makes hidden failures more likely. A strong CRA continuously updates site risk posture based on staff turnover, enrollment behavior, data quality, responsiveness, safety handling, deviation trends, and observed site judgment. That allows monitoring resources to follow risk instead of staying locked to a static schedule.
A fourth strategy is trend-focused communication. Average monitors write visit letters that describe issues. Strong monitors communicate risk patterns and operational meaning. They explain recurrence, consequence, urgency, and what good correction should look like. That clarity is essential because many sites fix findings superficially when the deeper workflow problem remains untouched. Strong CRA communication reduces that shallow response.
A fifth strategy is escalation discipline. Risk-based monitoring fails when the CRA identifies an important pattern but delays escalation out of optimism or discomfort. The monitor does not need to dramatize everything, but the monitor does need to know when a site issue has crossed from manageable imperfection into meaningful study risk. That judgment often defines the difference between a steady study and a messy rescue.
4. How CRAs Should Respond When Site Risk Starts Rising
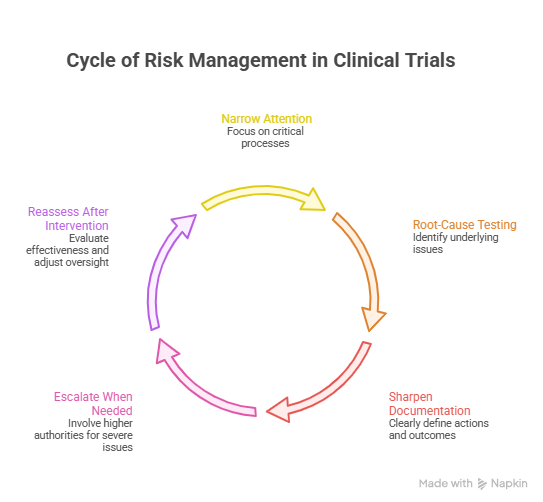
When site risk starts rising, the worst response is passive optimism. Many site failures become major only because weak signals were treated as temporary noise for too long. A strong CRA responds by tightening focus, clarifying the nature of the risk, and deciding what level of intervention fits the pattern.
The first response should be narrowing attention to the most consequential processes. If consent errors are emerging, consent control needs concentrated review. If AE delays are appearing, safety workflow and escalation timing must become immediate priorities. If endpoint-critical assessments look inconsistent, the CRA must stop diffusing attention across low-risk admin issues and move directly into the study’s most vulnerable areas. Risk-based monitoring always favors consequence over completeness theater.
The second response is root-cause testing. A finding may look like a training problem but actually be a staffing problem, a leadership problem, a process design problem, or a site culture problem. CRAs who treat every issue as retraining often get cosmetic improvement and real recurrence. The better question is what condition is allowing the error to repeat. Is the coordinator overloaded? Is backup staff unclear? Is the PI detached? Is delegation outdated? Is the site entering data too late to remember details accurately? Good risk response depends on honest diagnosis.
The third response is sharper documentation and follow-up. When risk is rising, vague monitor letters are not enough. The CRA should define what happened, why it matters, what corrective action is required, how recurrence will be prevented, and when proof of correction must be visible. That standard gets stronger when informed by clinical trial auditing & inspection readiness CRAs expert guide, handling clinical trial audits GCP preparation essentials, managing clinical trial documentation essential CRA techniques, managing protocol deviations GCP compliance strategies, and clinical trial auditing & inspection readiness CRAs expert guide. Clear documentation turns monitoring judgment into visible oversight.
The fourth response is escalation when the site’s control environment is weakening. A site with recurring critical findings, poor ownership, weak PI oversight, or rising safety/process instability may need sponsor visibility, more intensive monitoring, leadership involvement, or even enrollment restriction depending on severity. The CRA cannot solve every quality failure through encouragement alone.
The fifth response is reassessment after intervention. Risk-based monitoring is dynamic. A site that looked dangerous three weeks ago may stabilize with good action. Another site may look cooperative while deeper problems continue. Strong CRAs keep adjusting oversight level based on evidence, not on how reassuring the site sounds.
5. How To Build True CRA Mastery in Risk-Based Monitoring Over Time
CRA mastery in RBM grows from a specific kind of discipline: learning how studies actually fail. That sounds simple, but many monitors spend years reviewing findings without deeply studying patterns of failure. The best CRAs pay attention to what weak oversight looks like before inspection findings appear, what shallow CAPA sounds like before recurrence proves it weak, what stressed sites feel like before timelines break, and what kinds of “small” issues tend to precede serious quality consequences.
One major growth path is strengthening therapeutic understanding. When a CRA understands the disease area, visit logic, endpoint significance, safety sensitivity, and practical burden of the protocol, risk interpretation becomes much sharper. What looks like a minor schedule slip in one study may be critical in another. What looks like routine documentation noise in one protocol may directly affect evaluability in another. Better study literacy produces better monitoring choices.
Another growth path is improving central signal reading. Modern CRAs need to think beyond physical visits. Query aging, deviation trends, enrollment patterns, data anomalies, remote review alerts, turnaround time, and staff communication patterns all contribute to site risk posture. Monitors who only “become active” during visit windows will increasingly fall behind the way studies now operate. Remote awareness is no longer optional. It is part of baseline competence.
A third growth path is communication maturity. CRAs become more effective when they can influence sites without creating defensive shutdown. This takes precision. The monitor must be firm without being blurry, corrective without being theatrical, and specific enough that the site understands what must change in practice. Good communication is not softness. It is operational usefulness.
A fourth growth path is inspection-minded thinking. The best CRAs do not only ask whether something can be fixed. They ask how the pattern would look to a sponsor, auditor, inspector, or medical reviewer examining the site over time. That perspective sharpens prioritization fast. It forces the monitor to look at cumulative weakness, not just isolated paperwork.
The fifth growth path is broader trial literacy. A CRA who understands ICH guidelines simplified everything researchers need to know, phase I clinical trials explained objectives risks & process, phase II clinical trials goals examples and real-life insights, phase III clinical trials definitive guide & case studies, and phase IV clinical trials post-marketing studies clearly defined will monitor with more context and better judgment. Risk-based monitoring improves when the CRA sees the wider study logic, not just the immediate site tasks.
6. FAQs1
-
The goal is to focus monitoring attention where study risk is highest instead of treating all data, processes, and findings as equally important. It prioritizes patient safety, rights, critical data integrity, and high-consequence compliance areas.
-
It means reviewing smarter. Some low-value checks may receive less emphasis, but critical processes often receive more focused and more meaningful review. RBM is not lighter monitoring. It is more strategic monitoring.
-
High-value signals include consent problems, delayed AE or SAE handling, eligibility uncertainty, recurring protocol deviations, IP accountability issues, weak PI oversight, increasing query burden, slow query closure, endpoint inconsistency, and staff turnover that disrupts execution.
-
The CRA should assess consequence, pattern, recurrence, and control weakness. A serious issue usually affects patient safety, protocol compliance in critical processes, data tied to study endpoints, or the site’s ability to prevent the problem from recurring.
-
A finding is an observed issue. A risk signal is a clue that broader weakness may exist. One finding can be isolated. A risk signal points to a pattern, a vulnerable process, or a site condition that may generate future failures if left unchecked.
-
Because numbers alone do not tell the whole story. A site may appear responsive in metrics while deeper weaknesses exist in consent execution, late source entry, PI oversight, staff understanding, or superficial corrective actions. RBM still requires strong human judgment.
-
Study critical processes more deeply, read patterns instead of isolated errors, use central data proactively, strengthen root-cause thinking, improve escalation judgment, and learn how quality failures actually develop over time at sites.