Directory of Free Clinical Research Training Resources & Webinars (2026 Edition)
Free clinical research training can save money, but that is not the real reason it matters. The real advantage is speed. Good free resources let you close skill gaps before an interview, sharpen weak operational areas before a promotion cycle, and build technical language before you ever sit for a certification exam. In a field where employers quietly reject people for sounding vague, undertrained, or operationally soft, free training is not a side benefit. It is leverage.
This guide organizes the best no-cost clinical research learning resources into a practical career tool. It shows which free courses help beginners, which webinar libraries are strongest for regulation and trial operations, which resources are worth your time for CRC, CRA, pharmacovigilance, and data roles, and how to build a 90-day training stack that turns scattered learning into actual hiring value.
1. Why Free Clinical Research Training Matters More Than Most People Think
Most professionals underestimate how fast credibility forms in clinical research. It starts long before an employer checks your references. It shows up in how you talk about Good Clinical Practice, how precisely you discuss informed consent, whether you understand protocol deviations, and whether your answers reveal operational depth or just memorized buzzwords. That is why strong free training matters. It helps you stop sounding interested in clinical research and start sounding usable inside it.
The best free resources in 2026 are not random YouTube clips or recycled slide decks. The strongest options come from official or highly credible sources such as NIH, FDA, ClinicalTrials.gov, WHO-linked learning platforms, the Global Health Training Centre, MRCT Center, and TransCelerate, with many offering structured modules, archived recordings, downloadable materials, or certificates.
That matters because clinical research punishes shallow understanding. A coordinator who cannot explain adverse events, a monitor who cannot discuss clinical trial documentation under GCP, or a candidate targeting safety roles who cannot speak clearly about drug safety reporting timelines will sound expensive to train and risky to hire. Free training reduces that friction.
It also solves a second problem that hurts many job seekers: uneven knowledge. Someone may understand the clinical research coordinator role but be weak on case report forms. Another person may know the CRA career path but not the mechanics of site selection and qualification visits, audit readiness, or GCP compliance essentials for CRAs. Free resources let you tighten those weak links before they cost you an interview, a site visit, or a manager’s trust.
The smartest way to use free training is not to collect certificates. It is to match each resource to a career problem. If you are new, you need foundations. If you are interviewing, you need regulatory fluency and interview-ready terminology. If you are already working in research, you need sharper operational judgment, stronger cross-functional language, and niche depth in areas like pharmacovigilance case processing, signal detection, clinical research project planning, or clinical data systems.
2. Which Free Training Resources Are Best for Different Clinical Research Roles
A common mistake is using one training list for every role. That wastes time and creates false confidence. A future CRC does not need the same stack as a future CRA. Someone aiming for safety operations should not study like a site coordinator. Someone moving toward project management should not build a beginner-only curriculum and call it preparation.
If you want CRC or site-facing roles, prioritize resources that strengthen your operational reflexes. That means foundations in clinical trial protocol management, regulatory document management, essential adverse event reporting techniques for CRCs, GCP compliance strategies for coordinators, and patient recruitment mastery. For that path, the strongest free pairings are GHTC’s coordinator operations course, informed consent materials, NIH GCP frameworks, and ClinicalTrials.gov basics because those resources make you more useful at the site level.
If you want CRA or monitoring roles, focus on resources that improve inspection thinking, monitoring judgment, and guideline language. You want free training that helps you speak clearly about clinical research associate essential monitoring techniques, site selection and qualification visits, clinical trial auditing and inspection readiness, managing clinical trial documentation, and clinical trial monitoring terms every CRA should know. FDA clinical trial modules, MRCT’s ICH training, TransCelerate’s E6 resources, and GHTC’s E6(R3) course are stronger here than generic “clinical research intro” content.
If you want pharmacovigilance roles, free training should move you toward precision in case processing, signal detection, aggregate reports, regulatory submissions in pharmacovigilance, and risk management plans. FDA webinar resources and broader regulatory modules help more than generic academic content because safety hiring managers care about judgment, timelines, and regulatory language, not vague enthusiasm for “drug development.”
If you want data, TMF, or centralized operations roles, focus on resources that strengthen your understanding of electronic data capture systems, clinical trial management systems, clinical data management systems, remote patient monitoring tools, and electronic patient-reported outcomes tools. Free courses on governance, data sharing, registry literacy, and decentralized operations often deliver more value than another introductory GCP refresher.
If you want project management or leadership roles, use free webinars and frameworks to deepen your understanding of resource allocation, vendor management, stakeholder communication, budget oversight, and risk management in clinical trials. TransCelerate and MRCT are especially useful here because they expose you to design, implementation, inclusion, and cross-functional oversight issues that entry-level courses usually miss.
3. How to Separate High-Value Free Training From Low-Value Free Content
Free training becomes dangerous when it creates the illusion of competence. The wrong webinar makes people feel informed without making them more accurate, more compliant, or more operationally useful. That is why you need a filter.
First, ask whether the resource is close to the work. NIH, FDA, ClinicalTrials.gov, WHO-linked learning systems, MRCT, and Global Health Training Centre materials are valuable because they sit near regulation, execution, methods, or real trial operations. That proximity tends to produce better terminology, sharper expectations, and less fluff. The Global Health Training Centre, for example, explicitly positions its e-learning as peer-reviewed and regularly updated, while the FDA modules frame clinical-trial learning around current regulatory thinking, and MRCT’s ICH training focuses directly on interpreting and applying E6(R3).
Second, ask whether the resource teaches a skill or just a concept. A useful course helps you do something better. That may mean reading a protocol more intelligently, documenting consent more carefully, understanding serious adverse event reporting procedures, speaking more clearly about randomization, or catching the operational consequences of blinding choices. High-value free training improves judgment. Low-value free training just gives you vocabulary.
Third, check whether the resource reflects where the industry is moving. In 2026, training that ignores decentralized trial elements, patient-centered design, governance, data-sharing expectations, and guideline updates is already aging. That is why the newer WHO clinical-trial design course, MRCT decentralized-trial events, MRCT inclusion toolkits, and TransCelerate’s E6 and personalized-trials frameworks deserve attention. They help you think in the direction clinical operations is moving, not where it was five years ago.
Fourth, watch for certificate traps. A free certificate can help, but only when the learning behind it is real. Employers rarely care about a badge by itself. They care whether the training changed the way you speak, think, and solve problems. Someone who has worked through free resources and can intelligently explain ICH guidelines, institutional review board roles, IND applications, and sponsor responsibilities is far more credible than someone who only lists modules completed.
What is your biggest clinical research training problem right now?
Choose one. Your answer points to the fastest fix.
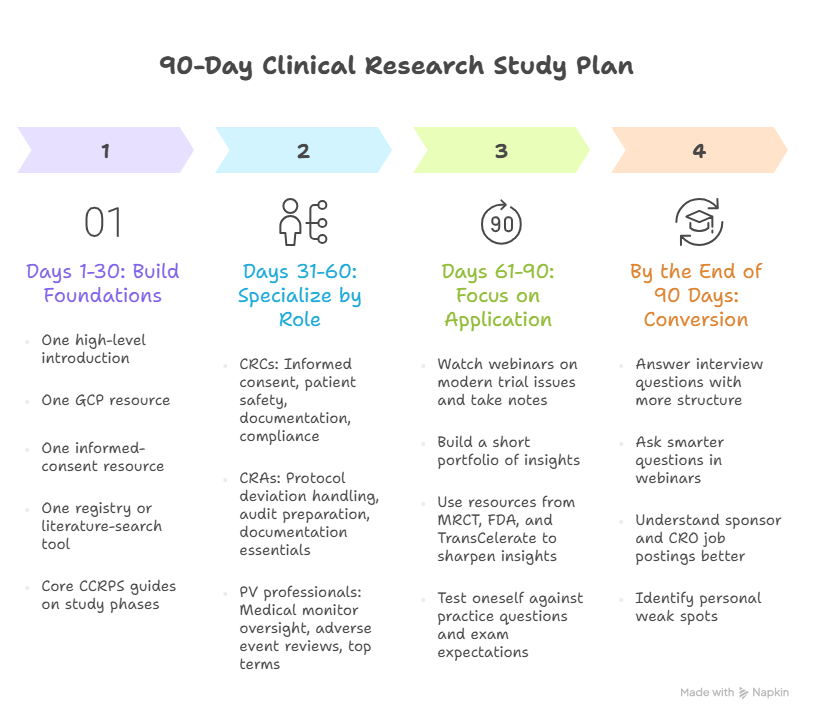
4. The Smartest Way to Build a Free 90-Day Clinical Research Study Plan
The best free training strategy is layered, not random. In the first 30 days, build foundations. That means one high-level introduction, one GCP resource, one informed-consent resource, and one registry or literature-search tool. A strong combination is GHTC Introduction to Clinical Research, GHTC or NIH/OBSSR GCP materials, GHTC consent learning, and ClinicalTrials.gov or NLM learning resources. This phase should also include reading core CCRPS guides on phase I, phase II, phase III, and phase IV studies so your terminology stops floating without structure.
In days 31 to 60, specialize by role. CRCs should move deeper into informed consent procedures, patient safety oversight, study documentation, and research compliance and ethics. CRAs should deepen protocol deviation handling, audit preparation, and clinical trial documentation essentials. PV professionals should target medical monitor oversight, managing adverse event reviews, and top terms for pharmacovigilance specialists.
In days 61 to 90, focus on application. Watch webinars that expose you to modern trial issues. Take notes in your own words. Build a short portfolio of insights: one page on consent risk points, one page on protocol deviation prevention, one page on adverse-event escalation logic, one page on how decentralized elements change oversight. Use resources from MRCT, FDA, and TransCelerate to sharpen those pages. Then test yourself against CRC exam practice questions, CRA certification practice work, CRC exam topic expectations, and common CRA certification mistakes.
The point of the plan is not just knowledge retention. It is conversion. By the end of 90 days, you should be able to answer interview questions with more structure, ask smarter questions in webinars, understand more of what sponsor and CRO job postings are actually demanding, and identify where your weak spots still live. That is where free training becomes career capital instead of content hoarding.
5. The Biggest Mistakes People Make With Free Clinical Research Training
The first mistake is confusing more content with more competence. Saving 40 webinar links and finishing none of them is not training. Watching hours of material without taking notes, testing recall, or connecting the lessons to clinical trial protocols, adverse event handling, clinical trial amendments, and research assistant data collection skills creates familiarity without execution value.
The second mistake is only studying what feels easy. Many learners stay inside intro courses because advanced topics expose what they do not know. That is exactly why you should lean into harder materials on governance, decentralized oversight, regulatory interpretation, inclusion, and data-sharing ethics. Those are the topics that differentiate serious candidates from people who only know definitions.
The third mistake is ignoring role context. Someone preparing for a site role who spends all their time on abstract policy webinars will sound detached from actual workflow. Someone aiming for sponsor or CRO roles who never studies implementation frameworks will sound operationally thin. The fix is to keep one eye on role-specific CCRPS guides and one eye on high-quality free external training.
The fourth mistake is failing to translate learning into visible proof. After finishing a course or webinar series, write three things: what changed in your understanding, what risk it helps you prevent, and how it affects your target role. That practice turns learning into language. Language turns into credibility. Credibility gets interviews.
The fifth mistake is assuming free means lower quality. Some free resources in clinical research are stronger than paid courses because they are closer to regulators, methods experts, or major trial-operating organizations. That does not mean every free resource is strong. It means you should evaluate on substance, not price.
6. FAQs About Free Clinical Research Training Resources & Webinars
-
They can be enough to strengthen your profile, improve interview performance, and close visible knowledge gaps. They are strongest when combined with practical experience, strong role positioning, and targeted study using clinical research certification providers comparisons, best clinical research certificate programs compared, and continuing education providers. Free training helps most when it makes you more precise, not when it becomes a substitute for real skill development.
-
A short introduction plus GCP plus informed consent is the strongest foundation. A beginner can start with the GHTC Introduction to Clinical Research, then add GCP learning and consent-focused training, while reading the CCRPS guides on clinical research terms, top CRC terms, and top CRA terms. That combination reduces confusion fast.
-
The strongest no-cost GCP options currently include NIH-linked materials, OBSSR’s social and behavioral GCP content, NIAID learning resources, GHTC’s ICH Good Clinical Practice E6(R3) course, and MRCT’s ICH training modules. They cover purpose, responsibilities, interpretation, and newer E6(R3) framing in ways that matter for current practice.
-
Both matter, but they do different jobs. Structured courses build foundations. Webinar archives expose you to current issues, implementation challenges, and emerging thinking. FDA, MRCT, and TransCelerate webinar archives are valuable because they often cover real-world operational and regulatory problems rather than just textbook definitions.
-
CRCs should prioritize coordinator operations, informed consent, GCP, registry literacy, patient recruitment, protocol management, documentation quality, and adverse-event reporting. The most useful pairings are GHTC coordinator-focused resources plus CCRPS articles on mastering patient recruitment, informed consent essentials for CRCs, protocol management, and essential AE reporting techniques.
-
CRAs benefit most from GCP interpretation, monitoring logic, documentation control, site-readiness thinking, protocol deviation management, and audit awareness. That makes FDA clinical trial training, MRCT ICH materials, TransCelerate E6 resources, and CCRPS guides on CRA monitoring techniques, inspection readiness, and essential CRA documentation techniques especially useful.