Best Oncology Clinical Research Conferences: Comprehensive Directory
Choosing the right oncology conference can strengthen trial judgment, sharpen operational instincts, and accelerate career growth, or it can drain budget and produce a notebook full of ideas that never improve recruitment, monitoring, safety, or execution. In oncology, the margin for shallow learning is small. Protocols are heavier, patient burden is higher, timelines are tighter, and scientific complexity punishes teams that stay generic. This directory is built to help professionals choose conference categories that actually improve site performance, oversight quality, trial delivery, and long-term oncology specialization.
1. Why Oncology Conferences Matter So Much in Clinical Research
Oncology conferences matter because cancer trials evolve fast, and that speed creates pressure on everyone touching the study. A site cannot recruit effectively when its team does not understand how a protocol fits into the current treatment landscape. A monitor cannot assess risk well when biomarker logic, line-of-therapy nuance, or emerging safety expectations remain fuzzy. A project manager cannot forecast burden correctly when real-world execution pressure is hidden behind polished protocol language. That is why the best conferences strengthen not only scientific awareness, but also clinical trial protocol management, patient recruitment, GCP compliance, patient safety oversight, and stakeholder communication.
The highest-value oncology conferences expose professionals to the decisions shaping tomorrow’s protocols before those decisions become everyday site burdens. That gives teams an edge. They begin to understand why inclusion criteria are tightening, why screening attrition is rising, why tissue requirements are becoming more demanding, why patient education must be more precise, and why safety narratives require stronger judgment. That kind of conference learning improves the work of clinical research coordinators, clinical research associates, professionals managing clinical trial documentation, teams responsible for serious adverse event reporting, and leaders overseeing clinical trial resource allocation.
They also matter because oncology punishes siloed professionals. A coordinator who only understands task flow but not therapeutic context will struggle when a patient asks why eligibility is so narrow. A CRA who knows checklists but not evolving oncology logic may miss which deviations truly matter. A project manager who tracks timelines but not the scientific drivers of complexity will underestimate risk. The right conference can close those gaps by connecting informed consent procedures, protocol deviations, adverse event handling, audit readiness, and regulatory responsibilities to the real scientific and operational pressures behind oncology research.
There is a career dimension too. Oncology is crowded with professionals who are smart, hardworking, and broadly competent. Broad competence alone rarely separates anyone. Conferences become career accelerators when they help a person become more specific, more credible, and more useful. A well-chosen conference can sharpen language for interviews, improve conversation quality with investigators and sponsors, deepen specialization, and create better follow-up opportunities through clinical research networking groups, LinkedIn groups for clinical research professionals, continuing education providers, clinical research staffing agencies, and clinical research job portals.
2. How To Choose the Right Oncology Conference for Your Role
The wrong way to choose a conference is by prestige, popularity, or fear of missing out. The right way is to choose based on the gap you need to close. If your biggest struggle is screening attrition, you need conferences that improve disease-setting understanding, biomarker logic, and patient eligibility conversations. If your struggle is protocol execution under pressure, you need meetings that improve clinical trial documentation, regulatory document management, protocol deviation prevention, GCP compliance for CRAs, and audit preparation. Prestige does not solve a role-specific weakness. Fit does.
For CRCs and other site-facing professionals, the best oncology conference is usually one that sharpens patient-facing judgment. That means better understanding of how burden feels to cancer patients, why visit schedules become overwhelming, how to explain procedures without flattening nuance, and how to catch confusion before it becomes noncompliance. Conferences with strong operational, supportive care, or disease-specific site content often produce more value than purely headline-driven scientific meetings. They make people better at patient recruitment, informed consent, adverse event reporting techniques for CRCs, CRC responsibilities, and time management for CRC exam-style prioritization.
For CRAs, value comes from conferences that improve oversight quality rather than generic exposure. Oncology monitoring requires better judgment around eligibility nuance, safety interpretation, source detail, site burden, and deviations that actually matter. The most useful conference tracks are often the ones that bring science and operations together. Those sessions help CRAs strengthen monitoring techniques, site selection and qualification thinking, investigator site management, documentation control, and inspection readiness.
Project managers, trial managers, data professionals, regulatory specialists, and safety teams should choose differently. PMs need conferences that improve forecasting, communication, vendor alignment, and execution realism. Data teams benefit from stronger understanding of endpoint logic and site-level data friction. Regulatory professionals should favor conferences that clarify oversight expectations and implementation consequences. Safety teams need meetings that help them interpret oncology harm signals in context. Those choices support clinical trial budget oversight, risk management in clinical trials, clinical data manager growth, regulatory affairs specialist development, and pharmacovigilance career advancement.2
3. Which Conference Categories Usually Deliver the Highest Return
The highest return usually comes from the conference category that sits closest to the pressure point hurting your current work. For many professionals, that is not the biggest conference in the field. It is the conference that answers the questions they face every week. A coordinator struggling with consent fatigue and patient burden will often gain more from a supportive-care or operational oncology meeting than from a prestige-heavy scientific congress. A CRA trying to improve oncology oversight may benefit more from sessions on eligibility complexity, source review, safety escalation, and execution failure patterns than from blockbuster efficacy headlines. This is where conferences start improving clinical trial protocol management, serious adverse event reporting procedures, case report form quality, protocol amendment handling, and quality assurance thinking.
Flagship congresses still matter. They are valuable for seeing the direction of travel across oncology. They help professionals understand which disease areas are heating up, which modalities are driving protocol complexity, where competition for patients is intensifying, and which trends may change site feasibility assumptions. That broad view helps teams think better about clinical trial sponsor responsibilities, clinical operations growth, clinical trial manager development, top clinical research organizations hiring, and major hospital research environments.
Disease-specific meetings often produce the clearest operational intelligence. They teach why a study is harder to recruit for, why tissue submission fails, why a certain patient population hesitates, why one line-of-therapy setting changes expectation management, or why a specific toxicity profile requires more proactive education. That level of context improves clinical research coordinator performance, clinical research assistant growth, research assistant compliance skills, laboratory coordination discipline, and patient safety oversight by investigators.
Operational, quality, and patient-centered meetings are often the most underrated category of all. They teach professionals where trials break in real life. They expose repeated failure modes: weak handoffs, unclear patient materials, burden-blind scheduling, delayed symptom escalation, site overload, inconsistent documentation, and amendment confusion. Those lessons are gold because they improve execution faster than generic inspiration ever will. That is where conferences start helping with clinical compliance officer judgment, clinical quality auditor development, vendor management, resource allocation mastery, and clinical research project manager growth.
4. How To Turn Conference Attendance Into Better Trial Performance
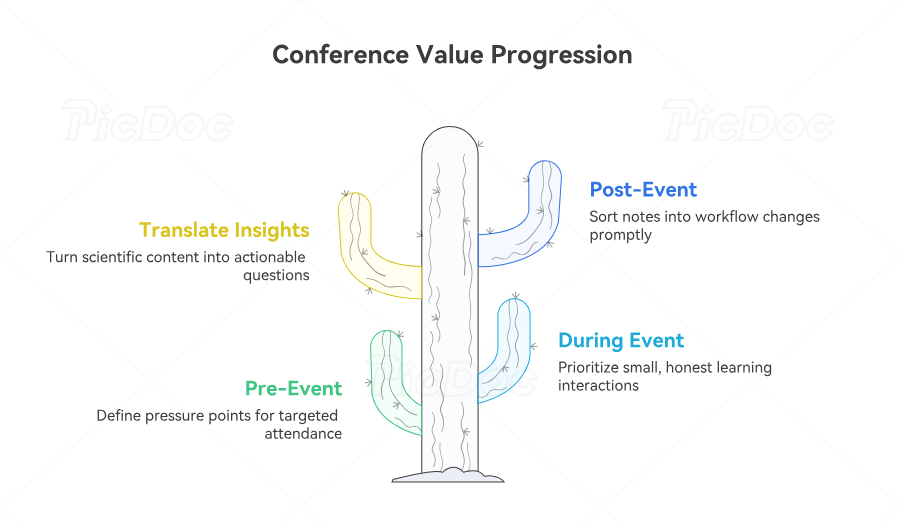
Conference value is determined before the event begins. The professionals who gain the most do not arrive looking for vague inspiration. They arrive with pressure points. A CRC might want better ways to explain oncology burden, prevent avoidable screen failures, and improve patient follow-through. A CRA may want stronger judgment around biomarker-driven eligibility, source review, safety escalation, and site support. A PM may want better forecasting for risk, vendor alignment, and feasibility. Those goals turn conference attendance into a tool for improving patient recruitment, informed consent procedures, clinical trial auditing readiness, budget oversight, and risk management.
During the conference, selective attention matters more than maximum attendance. Not every keynote deserves your time. Some of the most useful learning happens in smaller sessions, workshops, side conversations, poster discussions, and operational panels where people talk honestly about friction. That is where professionals often hear what actually goes wrong in oncology trials: how patients get lost between screening steps, why site burden is underestimated, where documentation drifts, why safety signals are reported late, and how protocol amendments create confusion. That kind of insight improves study documentation practices, regulatory document management, serious adverse event handling, clinical trial amendments control, and GCP training application.
The best attendees also translate scientific content into operational questions. If a session highlights a new biomarker dependency, the useful question is not only whether the science is exciting. The useful question is what that change means for tissue quality, turnaround time, screening failure, patient communication, budget, and site readiness. If a session highlights a new toxicity pattern, the real question is how patient education, call triage, escalation pathways, and documentation should change. This is where conferences start improving pharmacovigilance case processing, signal detection, risk management plans, medical monitor review, and clinical medical oversight.
After the conference, value decays fast unless learning is converted into action. Within days, notes should be sorted into workflow changes, leadership discussion points, follow-up contacts, further-reading needs, and risks that deserve early attention in active or upcoming studies. This is where exposure becomes advantage. Without that step, even a strong conference becomes expensive entertainment. With it, conference attendance can improve clinical data coordination, lead data analyst thinking, clinical trial assistant growth, clinical regulatory specialist development, and clinical research administrator leadership progression.
5. Common Mistakes That Waste Conference Budget and Learning Potential
The first mistake is choosing conferences that sound important instead of conferences that solve real problems. A giant conference can still be a weak investment if your role-specific needs are buried under content that never touches your daily work. Many professionals come back energized but unchanged because they consumed prestige, not precision. Precision is what improves clinical trial protocol management, deviation prevention, clinical trial documentation, audit readiness, and clinical compliance leadership.
The second mistake is attending without a learning framework. Passive note-taking creates the illusion of progress. It rarely produces better recruitment, safer reporting, stronger oversight, or clearer communication. Before the conference starts, a professional should know what they want to improve: science literacy, patient-facing conversations, monitoring quality, safety judgment, project execution, vendor evaluation, or career visibility. That structure helps transform conference content into better performance in roles such as clinical research associate, clinical research coordinator, regulatory affairs associate, quality assurance specialist, and clinical operations manager.
The third mistake is networking badly. Many professionals either avoid networking entirely or do it in a shallow, transactional way. They collect contacts without starting real conversations, then wonder why nothing develops afterward. High-value networking is selective and serious. It works best when you ask informed questions, speak concretely about the work, and follow up in a way that shows memory and substance. This supports movement into medical science liaison roles, stronger scientific communication, deeper KOL engagement, more credible medical monitor pathways, and better visibility through professional forums.
The fourth mistake is ignoring adjacent content. Oncology trials are never only about efficacy data. They are also about patient burden, documentation discipline, safety reflexes, regulatory clarity, staffing pressure, digital systems, and the operational cost of scientific ambition. Professionals who ignore supportive care, quality, regulatory, data, and patient-experience sessions often miss the exact content that would make them more effective. That broader lens strengthens research assistant performance, effective data collection and management, principal investigator team leadership, clinical trial protocol development awareness, and long-term specialization across the oncology research ecosystem.
6. FAQs
-
The best fit is usually a meeting that combines disease-specific oncology context with strong operational content. Coordinators gain the most when a conference improves how they explain burden, manage screening complexity, support patient follow-through, and prevent avoidable workflow breakdowns.
-
Yes, when the goal is to understand broad movement across oncology, competitive intensity, and future trial complexity. Their value drops when attendees expect them to solve role-specific operational problems without a clear session strategy.
-
CRAs should prioritize content that improves oversight quality: eligibility nuance, site burden, safety escalation, source expectations, biomarker-related workflow risk, and deviation patterns that matter clinically and operationally.
-
They help when they increase specificity. A strong conference improves the way you talk about the field, the kinds of people you can build relationships with, the problems you can solve better, and the signals you send in interviews and professional follow-up.
-
You return with pages of notes, but no clearer decisions, no practical workflow changes, no valuable new relationships, and no sharper understanding of your current studies or target role.
-
Project managers usually benefit most from conferences that connect science to execution. Pure science can be useful, but only when it helps forecast burden, improve risk planning, support vendors better, and strengthen communication with sites and sponsors.