Global Infectious Disease Clinical Research Sites: Interactive Directory
Infectious disease trials are where weak feasibility gets exposed fast. A site can look impressive on paper and still fail when specimen transport breaks, microbiology turnaround drifts, community trust is thin, follow-up collapses in stigmatized populations, or safety reporting slows outside business hours. That is why a real directory cannot just list hospitals, research centers, and countries.
This guide is built to help sponsors, CROs, CRAs, CRCs, and candidates evaluate infectious disease sites through the lens of operational fit. It connects site selection to the realities of clinical trial sponsor responsibilities, site qualification visits, GCP mastery, regulatory document management, and serious adverse event reporting so teams can choose sites based on execution, not reputation theater.
1. Why Infectious Disease Site Selection Fails More Often Than Teams Expect
Infectious disease feasibility goes wrong when teams confuse disease burden with research readiness. A region can have a massive patient load, but the wrong site inside that region can still underperform because screening is unstructured, consent is delayed, culture collection is inconsistent, coordinators are thinly stretched, pharmacy workflows are weak, or patient trust never becomes real engagement. Strong sites do not just see patients. They turn clinical burden into disciplined research through informed consent procedures, adverse event reporting techniques for CRCs, drug safety reporting timelines, protocol deviation control, and clinical trial documentation under GCP.
The other major mistake is treating infectious disease research like one category. It is not. A site that is excellent for vaccine trials may be weak in TB retention. A center that handles acute inpatient sepsis studies beautifully may struggle with HIV adherence, STI outreach, or long-tail viral hepatitis follow-up. A tropical disease institute may be exceptional for parasitic or arboviral studies but irrelevant for pragmatic respiratory infection protocols. That means site selection has to be driven by protocol design, endpoint demands, patient access pathways, and follow-up burden. Teams that understand primary versus secondary endpoints, randomization techniques, blinding in clinical trials, placebo-controlled trial design, and biostatistics in clinical trials make far better site decisions than teams that select on name recognition alone.
This topic also matters for careers because infectious disease research sharpens operational judgment fast. It teaches people how fragile recruitment really is, how trust affects retention, how safety reporting pressure changes behavior, and how protocol complexity shows up in real workflows. That is why professionals building toward the CRA career path, the CRC career path, the clinical research project manager path, the regulatory affairs specialist roadmap, and the quality assurance specialist roadmap should pay close attention to which site environments create strong operators and which ones hide weak process under big institutional names.
| Region / Site Type | Best-Fit Infectious Disease Programs | Strengths | Risks / Failure Modes | How To Use The Site Well |
|---|---|---|---|---|
| U.S. academic infectious disease referral center | Complicated infections, MDR organisms, hospitalized anti-infective studies | Specialist oversight, advanced diagnostics, strong safety judgment | Investigator overload, competing studies, slow internal routing | Use for medically complex protocols where nuance matters more than raw speed |
| U.S. HIV specialty clinic network | ART optimization, PrEP, long-horizon adherence studies | Patient trust, adherence experience, strong retention habits | Narrow eligibility if protocol ignores real treatment patterns | Best for trials where repeat contact and retention are decisive |
| U.S. vaccine research unit | Adult vaccine, pediatric vaccine, immunogenicity and safety studies | Visit-window discipline, startup speed, cold-chain reliability | Weak fit for severe inpatient infection studies | Choose for protocol-driven preventive research with dense schedules |
| U.S. sepsis / ICU tertiary hospital | Sepsis, hospital-acquired pneumonia, high-acuity anti-infective trials | Rapid case capture, specialist escalation, severe disease access | Off-hours screening gaps, consent timing failures | Activate only with aggressive around-the-clock screening coverage |
| U.S. STI and sexual health center | STI prevention, behavioral intervention, pragmatic sexual health studies | Community access, pragmatic recruitment, rapid identification | Retention loss if privacy concerns are underestimated | Design around convenience, trust, and stigma-aware communication |
| Canada microbiology-linked academic center | AMR, diagnostic validation, stewardship-linked anti-infective studies | Lab integration, source quality, disciplined oversight | Slower startup than aggressive sponsors want | Use when specimen integrity and data cleanliness matter most |
| U.K. NHS infectious disease teaching hospital | Pragmatic anti-infective studies, outpatient therapy, vaccine work | Structured pathways, guideline alignment, reliable protocol execution | System layers can slow approvals and contracting | Strong for multicenter studies with realistic startup planning |
| Western Europe travel medicine center | Imported infections, prophylaxis, traveler-related studies | Niche expertise, phenotype precision, globally aware investigators | Recruitment pools can be narrower than expected | Best for specialized protocols needing diagnostic certainty |
| Germany hospital epidemiology center | HAI, AMR, infection prevention trials | Methodical workflows, stewardship culture, procedural rigor | Can be rigid in community-facing pragmatic studies | Excellent for inpatient and systems-heavy protocols |
| Netherlands public health-linked site | Vaccine effectiveness, surveillance, longitudinal infectious disease follow-up | Data orderliness, registry logic, retention strength | Less useful for urgent acute recruitment | Use for follow-up-driven and public-health-linked studies |
| France tropical and imported disease unit | Parasitic disease, imported febrile illness, traveler cohorts | Specialist depth, diagnostic sophistication, global case mix | Volume may fluctuate sharply | Ideal for niche infectious disease studies needing expert confirmation |
| Spain HIV / hepatitis coinfection center | HIV, viral hepatitis, chronic infectious disease management studies | Long-term trust, specialty retention, adherence support | Weak fit for acute non-viral protocols | Use where chronic follow-up is central to endpoint quality |
| Italy pediatric infectious disease hospital | Pediatric vaccine, RSV, childhood respiratory infection studies | Pediatric expertise, family-centered workflows, careful safety oversight | Enrollment slows if caregiver burden is mishandled | Build around parent communication and visit convenience |
| Scandinavian registry-enabled site | Population-based vaccine and infectious disease follow-up studies | Longitudinal tracking, public-health linkage, retention quality | Less suited for urgent high-acuity enrollment | Choose for endpoint-driven long-horizon protocols |
| Eastern Europe hospital network | Respiratory infection, inpatient anti-infective, broad inclusion studies | Recruitment efficiency, motivated teams, predictable visit completion | Operational quality varies by center | Segment sites carefully instead of treating the region as uniform |
| Brazil urban infectious disease institute | HIV, dengue, arboviral disease, tropical infection studies | Disease diversity, experienced investigators, meaningful burden | Distance and income barriers can hurt retention | Pair with patient navigation and follow-up support |
| Mexico urban community-hospital hybrid | Respiratory infection, vaccine, STI, pragmatic anti-infective trials | Diverse populations, broad access, flexible recruitment channels | Higher education burden in mixed-literacy populations | Use simple consent language and strong coordinator coaching |
| Peru respiratory and tropical disease center | TB, respiratory infection, urban burden-driven studies | Relevant disease burden, local expertise, community engagement | Retention suffers without funded follow-up support | Strong for burden-heavy studies when retention is resourced properly |
| South Africa HIV and TB powerhouse site | HIV, TB, coinfection, prevention and treatment studies | Phenotype access, experienced teams, deep community credibility | Staff bandwidth can be strained by high study density | Best for complex longitudinal studies with serious sponsor support |
| Kenya community-linked infectious disease program | Malaria, maternal-child infectious disease, vaccine studies | Field engagement, local trust, burden relevance | Rural follow-up and specimen transport can be demanding | Use with strong field operations and transport planning |
| Uganda HIV cohort site | HIV prevention, treatment, viral suppression, adherence studies | Cohort continuity, counseling strength, trust-based retention | Complex protocols can outpace staffing if training is weak | Use for long-horizon HIV studies where retention is central |
| Nigeria tertiary infectious disease hospital | HIV, TB, viral outbreak preparedness, broad infectious disease studies | High-need populations, specialist depth, outbreak relevance | Infrastructure maturity differs sharply by center | Verify lab, pharmacy, and emergency escalation directly |
| North Africa urban fever / hepatology center | Hepatitis, febrile illness, hospital-linked infectious disease trials | Referral density, strong patient need, regional relevance | Administrative maturity varies by institution | Use only after real workflow validation during qualification |
| Middle East quaternary infectious disease hospital | Complex anti-infective, ICU infection, transplant-related infection studies | Advanced diagnostics, specialist depth, modern infrastructure | Recruitment pools may be narrower than forecast | Strong for high-complexity studies needing diagnostic confidence |
| Israel innovation-forward infectious disease center | Digital monitoring, vaccine, antiviral and diagnostics studies | Technical fluency, translational energy, agile investigators | Competition from multiple sponsors can dilute focus | Best for innovation-heavy protocols needing adaptable teams |
| India high-volume TB and respiratory center | TB, pneumonia, respiratory infectious disease studies | Huge patient flow, broad phenotype exposure, ambitious enrollment | Documentation quality varies by site tier | Segment site maturity carefully and monitor early |
| India vaccine and public health site | Pediatric vaccine, adult vaccine, prevention studies | Large reach, public-health linkage, operational ambition | Cold-chain and follow-up quality must be validated carefully | Use for scaled preventive studies with strong oversight |
| Pakistan tertiary infectious disease institute | Hepatitis, TB, respiratory and inpatient infectious disease studies | High burden, committed specialists, strong recruitment potential | Research staffing and infrastructure vary by center | Best with explicit training and source-document expectations |
| Singapore integrated national infectious disease center | Emerging pathogens, antiviral, vaccine, travel-linked infection studies | Organized systems, outbreak readiness, specialist coordination | Capacity can tighten for niche multinational programs | Excellent for high-discipline studies needing strong operational control |
| South Korea digital-health infectious disease site | Remote follow-up, respiratory infection, vaccine, digital symptom tracking | Digital fluency, strong hospital systems, innovation comfort | High protocol expectations expose weak sponsor planning fast | Best for tech-enabled studies with integrated data planning |
| Japan precision infectious disease center | Diagnostics, antiviral, hospital-linked infection studies | Methodical execution, source quality, careful follow-up | Recruitment can be slower for broad global timelines | Choose when exactness matters more than headline speed |
| Australia public health and outbreak network | Vaccine, outbreak response, respiratory infection, traveler studies | Governance strength, multicenter coordination, quality consistency | Geography can complicate dispersed follow-up | Use for coordinated multicenter studies with strong central control |
| Hybrid decentralized follow-up partner | Longitudinal safety, adherence, home symptom tracking, remote sample coordination | Patient convenience, retention support, reduced visit burden | Weak fit if high-acuity onsite diagnostics drive endpoints | Use as an extension layer, not a replacement for anchor sites |
2. How To Read This Directory Like A Sponsor, CRO, CRA, Or Candidate
Sponsors should use this directory as a feasibility filter, not a prestige list. The right question is not which site looks famous. The right question is which site type matches the trial’s most dangerous failure points. If your study depends on microbiology confirmation, lab timing, and specimen handling, then a site with weak laboratory coordination can destroy the program even if it recruits quickly. If the study depends on long-horizon adherence in a stigmatized population, then community trust matters as much as investigator expertise. Those decisions need to be anchored in clinical research project planning, resource allocation mastery, vendor management, risk management in clinical trials, and clinical trial budget oversight.
CRO teams and CRAs should read the directory as a monitoring forecast. Some infectious disease sites need heavy startup support but become stable after training. Others look polished during qualification and then drift later through slow query resolution, weak source notes, delayed safety escalation, inconsistent culture timing, or poor missed-visit recovery. The sites most likely to hold under pressure already show strength in CRA monitoring techniques, investigator site management, clinical trial documentation techniques for CRAs, GCP compliance for CRAs, and clinical trial auditing and inspection readiness. Infectious disease studies punish delay because a small workflow miss can damage culture timing, exposure interpretation, endpoint integrity, or patient safety narratives.
Candidates should read the directory as a map of where skills actually compound. HIV and TB sites build retention, counseling, adherence, and community-based follow-up judgment. Acute inpatient anti-infective sites sharpen case capture, screening speed, consent timing, and SAE reflexes. Vaccine sites teach schedule discipline, cold-chain thinking, and population-scale coordination. Public health-linked sites build longitudinal reasoning, registry awareness, and systems thinking. That is why the environments in this directory connect directly with the clinical research assistant roadmap, the clinical trial assistant career guide, the clinical compliance officer path, the clinical data coordinator career guide, and the clinical data manager roadmap. Once you understand site types, you stop applying blindly and start aiming at ecosystems where your skills will actually become valuable.
3. Regional Patterns That Actually Change Infectious Disease Feasibility
North America gives sponsors a wide spread of site models, from elite academic referral centers to community-facing sexual health clinics and dedicated vaccine units. The opportunity is range. The danger is false equivalence. A site that excels in resistant inpatient infections may be the wrong choice for preventive vaccine work. A high-acuity ICU center may struggle with the human work of long-horizon adherence. Teams that understand this usually also think more clearly about clinical trial protocol management, patient recruitment as a CRC skill, GCP compliance strategies for CRCs, research compliance and ethics, and laboratory best practices. They do not just ask how many patients a site sees. They ask how the site turns those patients into valid trial participants.
Western Europe and Canada often reward teams that care about systems discipline. Many sites there are strong in documentation, longitudinal logic, and structured workflows. The weakness is pace. Sponsors that expect aggressive startup without friction often misread the landscape and create their own delays. The best way to use these sites is to value them for control, not fantasy speed. That is why leaders who understand regulatory and ethical responsibilities for principal investigators, handling clinical trial audits, clinical regulatory specialist pathways, clinical quality auditor pathways, and continuing education providers in clinical research tend to run cleaner feasibility decisions across these geographies.
Africa, Latin America, South Asia, and parts of Southeast Asia are where lazy thinking creates expensive mistakes. Many sponsors reduce these regions to “high-enrollment” territory. That is a shallow read. The best sites in these regions often offer exactly what global studies need: meaningful disease burden, community-linked access, real-world diversity, strong investigator commitment, and deep experience with infectious disease realities that low-burden markets never see. The risk comes from assuming every site within a high-burden region shares the same operational maturity. Smart teams validate coordinator bandwidth, source structure, lab routing, pharmacy control, field follow-up, and adverse-event escalation directly. That is where knowledge of clinical research staffing agencies, job portals for clinical research careers, clinical research networking groups and forums, clinical research certification providers, and clinical research conferences and events becomes practical rather than abstract.
What is your biggest infectious disease site-selection blocker right now?
Choose one. Your answer points to the fastest operational fix.
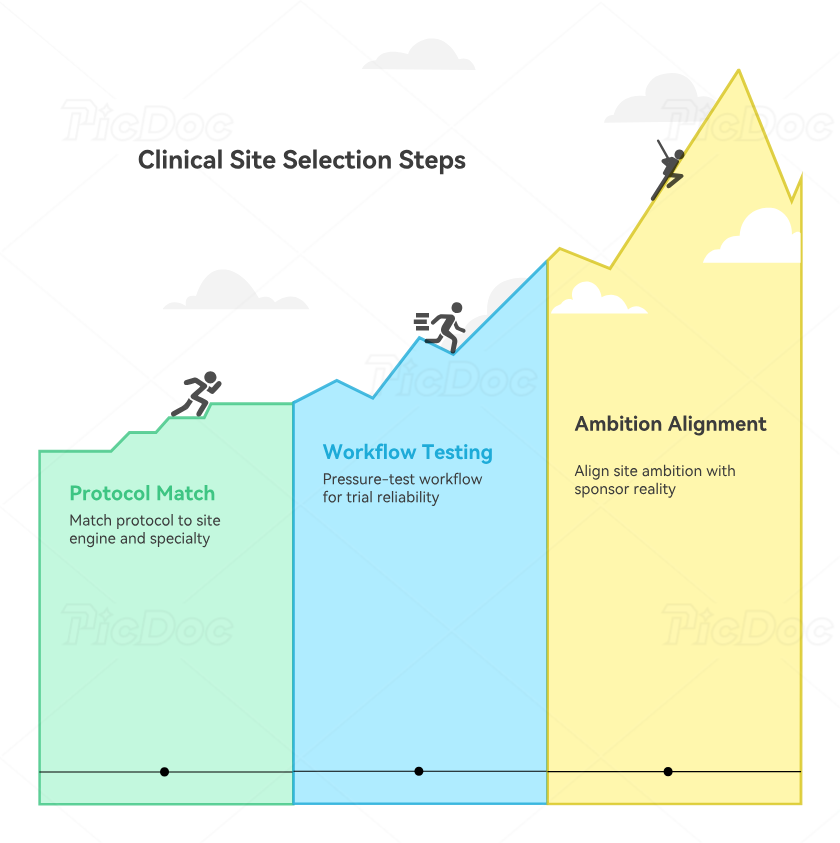
4. How To Match Infectious Disease Protocols To The Right Site Profile
The first step is matching the protocol to the site’s real engine. If the study is built around chronic retention, counseling, and stigma-sensitive populations, then HIV or hepatitis specialty networks often outperform famous acute-care hospitals. If the protocol depends on rapid inpatient identification, organism confirmation, and immediate treatment windows, then community outreach strength matters less than bedside screening execution. If the study is vaccine-heavy, then visit-window precision, cold-chain reliability, and community-scale scheduling matter more than tertiary-care prestige. That is why smart teams evaluate site fit alongside clinical trial volunteer registries, patient recruitment companies and tech solutions, hospitals and health systems running trials, academic medical centers with active trials, and CRO hiring ecosystems.
The second step is pressure-testing workflow maturity. Ask how the site identifies eligible patients, who owns prescreening, how specimens move, how missed visits are recovered, how AE and SAE reporting is escalated after hours, how protocol deviations are classified, and how coordinators cover each other when patient volume spikes. Infectious disease trials are unforgiving because timing matters. Delayed cultures, missed symptom windows, sloppy source notes, or inconsistent follow-up can destroy endpoint quality even when enrollment looks healthy. That is why serious selection teams study managing protocol deviations under GCP, pharmacovigilance case processing, signal detection and management, risk management plans in pharmacovigilance, and aggregate reports in pharmacovigilance before they finalize country and site mix.
The third step is matching site ambition to sponsor reality. A lean biotech cannot manage the same site portfolio as a global sponsor with mature regional infrastructure. Some sites deserve premium investment because they deliver high-complexity value. Others are ideal because they recruit steadily with manageable oversight. Great site strategy is portfolio design, not wishful selection. That is why experienced operators learn from the clinical operations manager roadmap, the clinical trial manager career guide, the medical monitor guide, the medical science liaison guide, and the principal investigator responsibilities guide. They understand that the wrong site mix does not just delay a study. It multiplies cost, weakens data credibility, and creates preventable safety stress.
5. Why This Directory Matters For Careers, Partnerships, And Execution Quality
For candidates, infectious disease research is one of the best training grounds in the industry because it forces clarity. You learn quickly whether you can organize messy screening pathways, communicate across stigma-sensitive populations, maintain source quality under pressure, and manage safety narratives without falling apart when timelines tighten. Those same skills transfer upward into project management, quality, pharmacovigilance, and medical oversight. That is why this directory has practical value for people using the ultimate CRC study guide, the CRC exam topics guide, the CRC exam practice test, the CRA exam mistakes guide, and the CRA practice test resource. The environments you choose early can shape whether you become merely experienced or genuinely sharp.
For sponsors and CROs, the value is even simpler. Good infectious disease site selection prevents expensive self-deception. It forces teams to stop asking whether a site is impressive and start asking whether the site can actually support the protocol’s hardest operational demands. The strongest studies are rarely built on the flashiest site list. They are built on sites that understand how patients move from identification to screening, consent, treatment, safety capture, retention, and closeout without process decay. That is where real trial quality lives.
This also matters for long-term partnerships. If a sponsor understands which site types fit which infectious disease programs, it can build smarter country strategies, more credible feasibility models, better training plans, and more stable site relationships. If a candidate understands which sites teach which muscles, career growth becomes intentional instead of random. That is why this topic connects so naturally with top clinical research journals and publications, LinkedIn groups for clinical research professionals, top freelance clinical research directories, remote CRA programs and jobs, and top trial sites and SMOs recruiting coordinators. People make better choices when they can finally see the ecosystem instead of isolated titles and site names.
6. FAQs About Global Infectious Disease Clinical Research Sites
-
The biggest mistake is treating disease burden like proof of site readiness. High patient volume means very little if screening pathways are weak, lab coordination is inconsistent, trust is low, or retention falls apart after enrollment. Good feasibility connects patient access to actual workflow ownership.
-
Sites with deep specialty clinics, counseling infrastructure, and trusted longitudinal relationships usually perform best. These trials live or die on retention quality, privacy sensitivity, and repeat engagement, not just physician prestige.
-
They are powerful only when operational maturity supports the burden. A high-burden site with weak source quality, fragile specimen routing, or poor follow-up can create more downstream damage than value. Burden helps. Controlled burden helps far more.
-
Ask how patients are identified, who owns screening, how specimens move, how missed visits are recovered, how after-hours safety escalation works, how stigma affects retention, and how many competing studies touch the same patient pool. Those answers matter more than generic enrollment promises.
-
Use it to identify which environment matches your strengths and the skills you want to build. Acute inpatient infectious disease sites sharpen speed and safety judgment. HIV and TB programs build retention and community trust. Vaccine units build schedule discipline and large-scale coordination.
-
Because outstanding clinical care and outstanding trial execution are different systems. Trials require research staffing depth, documentation discipline, specimen control, coordinator continuity, sponsor responsiveness, and long-horizon follow-up. Prestige may open the door, but process determines whether the study succeeds.