Leadership & Team Management: Clinical Trial PM Essentials
Clinical trial PM leadership becomes visible when the study starts resisting the plan: enrollment slows, vendors miss handoffs, sites need decisions, protocol amendments disrupt timelines, and leadership asks for confidence before the data supports confidence. Strong PMs do more than track tasks. They protect execution quality, decision speed, team accountability, and patient-safety alignment. The best leaders connect clinical trial project management, stakeholder communication, vendor oversight, and resource allocation into one operating system.
1. Lead the Trial Through Decisions, Not Status Updates
A clinical trial PM earns trust by turning scattered activity into clear decisions. Anyone can report that site activation is delayed, data queries are aging, monitoring visits are behind, or a vendor missed a deliverable. A leader explains what changed, what risk it creates, who owns the next move, what decision is needed, and how the team will know the issue is controlled. That mindset separates basic coordination from serious clinical trial manager leadership, clinical operations management, sponsor oversight, and GCP inspection readiness.
The painful failure in many trial teams is not laziness. It is decision fog. A CRA sees repeated source document gaps but frames it as a monitoring note instead of an escalation. A CRC sees recruitment friction but waits until enrollment is obviously behind. A vendor sees a data transfer defect but treats it as a technical inconvenience rather than a downstream database-lock risk. A PM has to convert those signals into operating decisions. That requires fluency in CRA responsibilities, CRC responsibilities, clinical data review, and risk-based monitoring.
Decision-led PMs run meetings differently. Every agenda item has a purpose: decide, escalate, unblock, confirm, or retire. Every risk has an owner and a next evidence point. Every overdue action has a business consequence attached to it. A weak meeting ends with “we will follow up.” A strong meeting ends with “the vendor will deliver the corrected transfer file by Thursday, data management will validate it by Friday, clinical operations will confirm whether any site-facing retraining is needed, and the PM will escalate if validation fails.” That is how stakeholder communication strategy, clinical trial documentation, trial safety monitoring, and protocol management become practical leadership tools.
| Leadership Situation | Common Failure Mode | Essential PM Move | Evidence of Strong Control |
|---|---|---|---|
| Site activation delay | Team reports delay without naming the blocker | Separate contracting, regulatory, training, budget, and feasibility barriers | Activation tracker connected to start-up checklist controls |
| Enrollment underperformance | PM waits until missed target becomes unrecoverable | Trigger rescue planning by site, indication, and patient pathway | Recruitment action log tied to patient recruitment trends |
| Site staff turnover | Knowledge transfer stays informal | Force role-based retraining and responsibility confirmation | Training evidence aligned with GCP training requirements |
| Vendor milestone slippage | Vendor gives optimistic recovery language without proof | Require dated recovery plan, owner map, and escalation path | Governance minutes from vendor management controls |
| Data query aging | Queries treated as data team problem only | Connect site behavior, monitor follow-up, and database-lock impact | Query trends reviewed through clinical data verification |
| Protocol deviation spike | Team counts deviations but misses the pattern | Classify root cause by procedure, training, feasibility, or oversight | CAPA linked to protocol deviation corrective actions |
| Safety reporting confusion | Clinical and safety teams disagree on clock start | Clarify first awareness, seriousness, escalation, and submission pathway | Process proof aligned with drug safety timelines |
| CRA workload imbalance | High-risk sites receive the same attention as stable sites | Rebalance monitoring effort by risk, volume, and issue history | Plan supported by risk-based monitoring strategy |
| Leadership pressure for faster timelines | PM accepts speed without naming quality tradeoffs | Present options with risk, cost, quality, and resourcing impact | Decision record tied to resource allocation mastery |
| Remote monitoring friction | Remote review creates silent evidence gaps | Define document access, source review limits, and escalation rules | Model based on remote monitoring techniques |
| Investigator meeting weakness | Meeting becomes slide delivery instead of behavior alignment | Focus sites on preventable errors, escalation standards, and protocol pressure points | Agenda linked to investigator meeting strategies |
| Budget overrun | Team notices spend after variance becomes political | Track burn rate against milestone value, change orders, and vendor output | Budget log informed by trial budget management |
| Stakeholder misalignment | Functions optimize their own deadlines | Translate each function’s concern into trial-level consequence | Decision notes from stakeholder communication planning |
| Inspection anxiety | Team starts organizing evidence after notice arrives | Maintain inspection-ready proof chains throughout execution | Readiness file aligned with CRA inspection readiness |
| Principal investigator disengagement | PI involvement becomes signature-based | Escalate oversight expectations through documented governance | PI evidence tied to PI regulatory responsibilities |
| Consent process errors | Team treats consent deviations as isolated site mistakes | Review training, version control, delegation, and screening workflow | Controls mapped to informed consent compliance |
| Retention decline | Retention receives attention only after dropout | Track visit burden, patient communication, and site follow-up quality | Retention plan built from patient retention strategies |
| Regulatory document gaps | Missing documents discovered near milestone review | Assign document ownership, review rhythm, and escalation rules | TMF support from regulatory document management |
| Amendment disruption | Amendment launched without impact mapping | Assess training, consent, budget, vendors, systems, and site burden | Change plan linked to clinical trial amendments |
| Medical monitor bottleneck | Medical decisions sit outside project visibility | Create escalation rules for safety, eligibility, and clinical judgment delays | Review pathway tied to medical monitor AE review |
| Quality issue recurrence | CAPA closes after retraining only | Test whether recurrence stopped with objective evidence | Effectiveness checks aligned with quality auditor discipline |
| Team conflict | PM avoids tension until work slows | Separate facts, ownership, decision rights, and escalation history | Conflict notes connected to PM communication strategy |
| Unclear accountability | Everyone knows the task, nobody owns the outcome | Define accountable owner, contributors, deadline, and acceptance criteria | RACI discipline supporting clinical research PM career skills |
| Technology adoption issues | Team blames the system while process defects remain hidden | Separate user training, configuration, data quality, and vendor support issues | System plan informed by clinical research technology adoption |
| Database lock risk | Lock preparation begins after the team already knows data are messy | Drive query closure, SDV completion, coding review, and reconciliation early | Lock readiness supported by data review skills |
| Executive reporting | Slides look clean but hide unresolved decisions | Report risk, options, decision need, and consequence of delay | Leadership pack tied to trial manager leadership |
| Team burnout | PM pushes harder without redesigning workload | Reduce noise, prioritize critical path, and escalate capacity gaps | Resourcing evidence connected to clinical trial resource allocation |
2. Build a Team Operating System That Makes Accountability Visible
Strong clinical trial teams do not rely on personality, memory, or heroic effort. They rely on an operating system. That system should define decision rights, escalation routes, meeting rhythms, risk thresholds, deliverable owners, quality expectations, vendor obligations, site support standards, and executive reporting rules. The PM’s job is to make accountability visible before the trial starts drifting. This is where clinical research PM development, clinical trial manager advancement, operations manager growth, and sponsor responsibilities become day-to-day management behaviors.
The most useful team structure starts with a responsibility map. For each critical process, define the accountable owner, supporting functions, required evidence, escalation trigger, and expected decision time. Enrollment belongs to clinical operations, but feasibility, contracts, patient materials, site engagement, medical eligibility questions, and recruitment vendors all influence enrollment. Data quality belongs to data management, but CRAs, sites, EDC setup, protocol design, query wording, and training quality all affect it. Safety belongs to pharmacovigilance, but sites, investigators, medical monitors, and clinical operations can break safety timelines. A PM who understands pharmacovigilance essentials, case report form best practices, primary and secondary endpoints, and protocol deviation control can lead across those seams.
Accountability also requires fewer, sharper meetings. A clinical trial can drown in recurring calls that create conversation without control. The PM should separate tactical execution meetings, vendor governance meetings, risk review meetings, data review meetings, safety review touchpoints, site activation reviews, and executive steering updates. Each meeting needs a different output. Tactical meetings should unblock work. Governance meetings should measure performance. Risk reviews should challenge assumptions. Executive meetings should force decisions. When every meeting has the same format, the team loses urgency. Strong PMs use stakeholder communication, vendor governance, risk-based monitoring, and GCP audit preparation to keep meeting architecture tied to trial risk.
3. Manage Performance Through Risk, Evidence, and Escalation Timing
Clinical trial PMs often lose control because they escalate too late or escalate too vaguely. “The site is slow,” “the vendor is behind,” or “data quality is poor” does not help leadership decide. A strong escalation names the metric, trend, root cause hypothesis, impact, options, recommended action, decision owner, and deadline. For example, enrollment risk should include site-level screening volume, screen failure reasons, activation age, referral source performance, patient burden, competing studies, and recovery forecast. That level of leadership uses patient recruitment insights, site monitoring strategies, patient retention techniques, and clinical trial cost estimating as decision support.
Performance management should protect the critical path. A PM should know which workstream can slip without damaging the study and which one silently threatens the whole trial. Delayed training may block activation. Delayed monitoring may hide deviation patterns. Delayed data cleaning may threaten interim analysis. Delayed safety reconciliation may create inspection exposure. Delayed vendor testing may compromise randomization, drug supply, or EDC reliability. The PM’s leadership value is the ability to distinguish noise from threat. That requires literacy across randomization techniques, blinding in clinical trials, data monitoring committee roles, and clinical trial safety monitoring.
The best PMs also manage tone. They create urgency without panic, accountability without blame, and transparency without chaos. Teams hide risk when every escalation becomes punishment. Teams also drift when every excuse is accepted. The leadership balance is firm, specific, and evidence-based: “This issue is late, this is the patient or quality impact, this is the owner, this is the recovery date, and this is when we escalate again.” That approach strengthens clinical trial PM communication, clinical operations leadership, clinical compliance officer skills, and quality assurance career growth.
What is your biggest clinical trial team management blocker right now?
Choose one. Your answer points to the leadership system that needs the fastest repair.
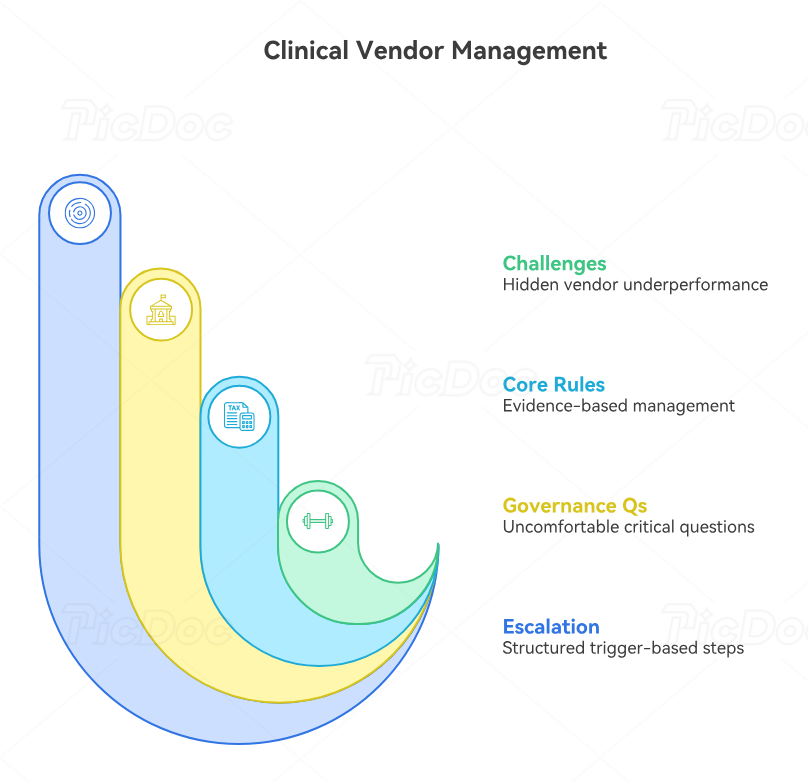
4. Lead Vendors Like Extensions of the Trial Team
Vendor leadership is one of the hardest parts of clinical trial PM work because vendors can appear controlled on slides while quietly weakening the study. A vendor can be “green” on status while deliverables lack quality, recovery dates keep moving, data transfers require rework, monitoring reports arrive late, or helpdesk issues frustrate sites. A PM should manage vendors through evidence, not optimism. That means clear scope, service levels, deliverable acceptance criteria, risk thresholds, governance cadence, escalation rules, and documented decisions. This is the heart of clinical trial vendor management, sponsor oversight, resource allocation, and project manager career advancement.
The best vendor governance questions are uncomfortable but necessary. What deliverables are late? What deliverables were accepted only after rework? What downstream teams are carrying vendor defects? What site complaints are repeating? What risks have stayed open for more than two governance cycles? Which KPIs measure activity but miss quality? Which change orders reflect scope growth, and which reflect weak planning? These questions protect the trial from polite underperformance. They also make the PM a stronger bridge between clinical operations, clinical data management, clinical safety monitoring, and regulatory compliance.
Vendor escalation should never feel random. The PM should define what triggers escalation before the problem happens: missed milestone, unresolved issue age, repeated rework, unresolved site-impacting defect, safety-risk delay, audit-relevant documentation gap, or leadership decision requirement. Escalation should ask for a decision, correction, resource, or accountability change. Weak escalation only says “this is concerning.” Strong escalation says “the EDC vendor has missed two validated transfer dates, database lock impact begins on this date, the recommended option is temporary daily defect triage with sponsor data management approval authority, and executive support is needed if the next validation fails.” That level of clarity supports clinical trial data review, audit readiness, remote monitoring control, and clinical quality auditing.
5. Protect Team Culture by Removing Ambiguity, Bottlenecks, and Hidden Work
Clinical trial teams burn out when the work is urgent, ambiguous, and invisible at the same time. The PM cannot remove every deadline, but the PM can remove confusion. People need to know which work matters most, which issues deserve escalation, which decisions are frozen, which tasks can wait, and which deliverables protect patient safety or regulatory credibility. Strong leaders reduce hidden work by making workload visible across CRAs, CRCs, data managers, safety teams, regulatory leads, medical monitors, and vendors. This connects directly to CRA career skills, CRC responsibilities, medical monitor review, and clinical regulatory specialist skills.
The leadership technique that matters most is prioritization with consequence. A PM should rank work by patient safety, regulatory risk, critical path, data integrity, site burden, cost exposure, and executive dependency. When everything is urgent, teams start making private decisions about what to ignore. That is dangerous because the ignored item may be the one that creates the inspection finding, missed enrollment target, database-lock delay, or safety reporting exposure. The PM has to make tradeoffs visible and documented. This is why GCP compliance, inspection readiness, protocol management, and patient safety oversight must sit inside project leadership.
Culture also improves when the PM creates psychological safety around early risk disclosure. Teams should feel rewarded for surfacing problems while they are still fixable. A CRA who identifies a pattern early should not feel like they created trouble. A site that admits workflow confusion should not be punished into silence. A vendor that flags a defect early should be held accountable, but the transparency should be valued. The PM’s standard should be simple: early bad news is useful; late bad news is expensive. That leadership style supports better site monitoring visits, patient retention planning, adverse event reporting, and clinical trial safety monitoring.
6. FAQs: Leadership & Team Management for Clinical Trial PMs
-
The most important skill is decision leadership. A clinical trial PM must turn activity, risk, delays, and cross-functional friction into clear decisions with owners, timelines, and evidence. Technical knowledge matters, but the PM’s value comes from connecting people and consequences. A strong PM understands clinical trial project management, stakeholder communication, resource allocation, and vendor management, then uses that knowledge to keep the trial moving without hiding quality or safety risk.
-
A PM improves accountability by defining owners, acceptance criteria, deadlines, escalation triggers, and evidence expectations before work begins. “Follow up with the site” is weak accountability. “CRA to confirm corrected delegation log by Friday, upload evidence to the TMF, and escalate if PI signature remains missing” is stronger. Accountability should be visible in action logs, risk registers, meeting minutes, and governance decisions. This approach supports regulatory document control, CRA documentation techniques, GCP compliance, and clinical quality auditing.
-
The PM should translate conflict into trial impact. Instead of letting each function defend its own workload, the PM should ask what the disagreement means for patient safety, data integrity, timelines, inspection readiness, cost, and site burden. Then the PM should document options, tradeoffs, and the needed decision. This prevents personality conflict from replacing project logic. Strong conflict handling depends on stakeholder communication strategy, clinical data verification, drug safety reporting awareness, and vendor oversight.
-
Senior leadership needs clarity, not noise. An effective PM presents the current risk, evidence behind the risk, decision required, options available, recommended action, and consequence of delay. Leadership updates should never bury unresolved decisions under attractive progress slides. A strong PM can explain whether a timeline is recoverable, whether additional resources are justified, whether vendor escalation is needed, and whether quality risk is increasing. This skill grows through clinical trial manager career development, clinical operations leadership, resource allocation planning, and sponsor responsibility awareness.
-
A PM prevents vendor problems by managing scope, service levels, quality expectations, issue aging, governance cadence, escalation triggers, and acceptance criteria from the beginning. Vendor performance should be measured by useful outputs, not polished updates. The PM should track missed deliverables, rework, site complaints, data defects, support delays, and unresolved risks. Vendor leadership should connect directly to vendor management essentials, clinical trial technology adoption, clinical data management, and inspection readiness.
-
A PM keeps the team motivated by reducing ambiguity, protecting priorities, recognizing early risk reporting, and making workload visible. Motivation drops when people feel overloaded, blamed, or unclear about what matters most. The PM should separate urgent from important, remove unnecessary meetings, escalate capacity gaps, and show how each role protects patient safety, data quality, and regulatory credibility. This leadership style supports patient safety oversight, site monitoring quality, patient retention, and GCP training discipline.