Top Global Neurology Clinical Trial Sites: Directory & Capabilities
Neurology trials punish weak site strategy faster than many other therapeutic areas. Recruitment moves slower, endpoint variability is harder to control, caregiver burden changes retention, imaging and cognitive workflows must stay precise, and protocol complexity can overwhelm sites that look strong on paper. This directory is built to solve that problem. It shows which neurology site profiles deserve priority, which capabilities actually matter in 2026, where sponsors and CROs get misled, and how to align site selection with enrollment, data quality, safety, and long-term execution strength.
1. Why Neurology Trial Site Selection Breaks More Studies Than Most Sponsors Expect
A neurology study can have a strong protocol, a solid budget, and a credible sponsor, yet still stall because the site mix was wrong from day one. That usually happens when teams overvalue brand-name institutions and undervalue operational fit. A site may have a famous neurology department but weak coordinator bandwidth, slow startup, poor caregiver communication, inconsistent cognitive testing discipline, or limited experience handling the exact endpoint structure your protocol requires. That is why smart teams pair site selection and qualification visits with stronger investigator site management, tighter clinical trial sponsor responsibilities, clearer clinical research project planning, and more disciplined risk management in clinical trials.
Neurology also exposes a deeper truth about site quality: the right site is rarely defined by PI prestige alone. It is defined by the whole operating system around the PI. Can the site recruit from the correct patient pool instead of a vaguely related neurology population? Can it manage caregiver-heavy visits? Can it maintain endpoint consistency across raters? Can it document deviations before they metastasize into inspection pain? Can it escalate worsening symptoms, falls, hospitalizations, or neurological adverse events through strong adverse event reporting techniques, defensible serious adverse event procedures, consistent GCP compliance strategies for coordinators, and clean clinical trial documentation under GCP?
Another major failure point is indication mismatch. Sponsors sometimes choose “top neurology sites” as if Alzheimer’s disease, Parkinson’s disease, epilepsy, multiple sclerosis, stroke recovery, migraine, ALS, neuromuscular disease, and rare neurogenetic disorders all reward the same site model. They do not. A site that excels in movement disorders may struggle with large memory-disorder screening funnels. A stroke center may be excellent at acute workflows but weak for long-duration cognition-heavy retention. A high-enrolling MS site may still have limited value if your protocol depends on advanced imaging harmonization, infusion logistics, or digital biomarker compliance. That is why strong teams connect patient recruitment strategy, realistic clinical trial protocol management, clearer protocol deviation handling, sound primary versus secondary endpoint planning, and disciplined clinical trial resource allocation before the first site contract is even finalized.
| Site Profile | Best For | Core Capabilities | Main Risk / Failure Mode | Best Sponsor Question |
|---|---|---|---|---|
| US academic memory disorders center | Alzheimer’s and MCI studies | Cognitive testing, biomarker workups, caregiver-heavy visits | Slow startup and PI overextension | How many screen-ready patients convert monthly? |
| US community memory clinic network | Broader dementia recruitment | Large referral base, practical retention workflows | Variable rater consistency across sites | How is endpoint standardization protected? |
| US movement disorders academic unit | Parkinson’s and atypical parkinsonism | Experienced raters, device familiarity, deep specialist referrals | Competing studies dilute enrollment | What is your current competing-trial burden? |
| US epilepsy referral center | Drug-resistant epilepsy trials | Seizure characterization, EEG access, complex medication histories | High screen failure if inclusion criteria are narrow | How do you prescreen before formal consent? |
| US MS infusion-capable center | Relapsing and progressive MS studies | Imaging coordination, infusion logistics, relapse assessment | Operational strain during heavy infusion schedules | Can trial infusions fit into real scheduling capacity? |
| US stroke recovery rehabilitation site | Post-stroke rehab and device studies | Functional endpoints, rehab integration, caregiver coordination | Retention drops in long follow-up designs | How do you retain mobility-limited subjects? |
| US neuromuscular disease center | ALS, SMA, muscular dystrophy | Rare disease recruitment, respiratory monitoring, caregiver-intensive support | Small pools limit backup options | Which rare-disease registries feed your site? |
| US headache and migraine specialty clinic | Migraine prevention and acute treatment trials | Large outpatient volumes, diary adherence coaching | Weak diary compliance undermines data | How do you rescue poor ePRO adherence? |
| Canadian academic cognitive neurology center | Dementia and neurodegeneration | Structured assessments, bilingual support, strong academic rigor | Long internal approvals | What is your realistic activation timeline? |
| Canadian MS specialty center | MS and neuroimmunology | Imaging access, relapse monitoring, long-term patient relationships | Enrollment caps from competing protocols | What percentage of your eligible pool is already committed? |
| UK NHS cognitive disorders site | Alzheimer’s, vascular dementia | Integrated referral pathways, strong documentation culture | Resource pressure limits coordinator bandwidth | Who actually owns day-to-day recruitment operations? |
| UK movement disorders unit | Parkinson’s studies | Experienced clinicians, strong patient follow-up systems | Visit windows get tight in overloaded clinics | How are protocol visits protected from clinic overflow? |
| Germany university neurology center | Complex biomarker and imaging trials | High technical rigor, advanced MRI and lab infrastructure | Operational flexibility can be limited | How quickly can amendment-driven changes be implemented? |
| Germany stroke network hospital site | Stroke and neurovascular studies | Acute workflows, imaging readiness, multidisciplinary coordination | Lower strength for long-term cognition retention | What long-horizon retention support exists after acute care? |
| France academic neurodegeneration site | Memory and rare neuro studies | Specialist depth, biomarker sophistication, academic credibility | PI-driven sites can bottleneck decisions | How much operational authority sits below the PI? |
| Spain hospital-based neurology research unit | Parkinson’s, migraine, MS | Strong patient relationships, balanced academic and operational workflows | Variable regional startup timelines | Which approvals historically delay activation most? |
| Italy movement disorders referral site | Parkinson’s and device-assisted care studies | Deep phenotype knowledge, strong specialist follow-up | Caregiver scheduling friction can slow visits | How do you accommodate caregiver-dependent attendance? |
| Netherlands cognitive trial center | Cognition-heavy and digital biomarker studies | Protocol discipline, digital tool adoption, strong data quality culture | Smaller populations constrain rapid rescue enrollment | How expandable is your referral network? |
| Belgium CNS hospital research site | Mixed neurology portfolios | Good multilingual operations, efficient startup teams | Portfolio spread can dilute focus | Which neurology indications are true priorities here? |
| Sweden neuroimmunology center | MS and autoimmune neurology | Longitudinal disease management, disciplined documentation | Limited volume for broad recruitment-heavy trials | Do you fit rare-depth protocols or mass-enrollment protocols better? |
| Denmark headache research clinic | Migraine and outpatient neuro studies | High diary engagement, organized patient communication | May be less suited to highly invasive designs | What procedure burden is realistic for your population? |
| Australia academic neurology network site | Parkinson’s, MS, dementia | Strong English-language operations, reliable documentation, academic depth | Geographic spread complicates retention for some cohorts | How far do participants travel for repeat visits? |
| Japan movement disorders and dementia center | Parkinson’s and dementia trials | High procedural rigor, specialist assessment discipline | Conservative timelines for protocol change adoption | How are retraining and change control executed? |
| South Korea university neurology site | Imaging-rich and device-enabled neuro trials | Advanced technology workflows, efficient hospital systems | Feasibility optimism can exceed real eligible pools | What percentage of projected patients met criteria in prior studies? |
| Singapore tertiary neuroscience center | High-complexity regional hub studies | Multidisciplinary coordination, strong technical infrastructure | Smaller catchment size for very rare cohorts | Which cross-border referral streams are active? |
| India high-volume neurology hospital research unit | Large recruitment targets and mixed neurology portfolios | High patient throughput, growing research capacity | Data quality can vary sharply by site leadership strength | How stable is the coordinator and rater team? |
| Brazil academic neurology referral center | Movement disorders, epilepsy, stroke follow-up | Large urban referral pools, strong specialist case mix | Retention and logistics need active operational support | What transportation and follow-up support exists? |
| Israel specialist neuroscience research site | Biomarker-heavy and innovation-forward studies | Scientifically sophisticated teams, fast technical adaptation | Limited scale for very broad recruitment needs | Is this site a depth play or volume play? |
2. The Top Neurology Site Capability Models Ranked for 2026
10. High-Volume Headache and Migraine Specialty Clinics
These sites deserve more attention than many sponsors give them because migraine studies live or die on outpatient flow, diary adherence, recurring visit discipline, and patient communication quality. A glamorous academic name is less valuable here than a site that can manage large screening funnels, teach participants how to complete digital reporting correctly, and protect primary endpoint integrity through strong case report form discipline, cleaner data management workflows, tighter patient recruitment execution, and fewer avoidable protocol deviations.
9. Stroke Recovery and Neurorehabilitation Sites
These sites matter because stroke studies often demand functional recovery assessment, therapy coordination, mobility-sensitive scheduling, and careful long-term follow-up. Teams that choose them well benefit from stronger patient safety oversight, clearer stakeholder communication, better resource allocation, and more realistic clinical trial budget oversight. Their main weakness appears when acute-care strength gets mistaken for strong chronic retention performance.
8. Epilepsy Referral Centers
Epilepsy sites rank highly because seizure characterization, medication complexity, caregiver reporting, and safety escalation require unusually disciplined teams. Strong epilepsy centers bring depth in classification, access to specialist populations, and better screening accuracy. The best ones combine tight adverse event handling, defensible drug safety reporting timelines, cleaner clinical trial monitoring techniques, and reliable research compliance and ethics mastery. Their common failure mode is overestimating how many “epilepsy patients” are actually protocol-fit.
7. MS Specialty Centers With Imaging and Infusion Strength
Multiple sclerosis studies reward sites that can harmonize imaging, track relapses correctly, coordinate infusion logistics, and retain patients through long timelines. A strong MS site supports better biostatistical planning, cleaner sample size assumptions, steadier clinical data manager workflows, and tighter vendor management when imaging and central reads matter. Their risk emerges when technical capacity looks impressive but operational capacity is already saturated.
6. Rare Neuromuscular Disease Centers
Rare neurology centers sit high in this ranking because they deliver what mass-market sites cannot: concentrated expertise, caregiver infrastructure, phenotype accuracy, and access to tightly defined populations. That makes them invaluable for ALS, SMA, muscular dystrophies, and related protocols. Their success depends on stronger volunteer registry use, smarter staffing strategy, good continuing education pipelines, and precise principal investigator responsibilities. Their weakness is scale. They are depth plays, not volume miracles.
5. Community-Connected Memory Clinic Networks
These sites matter because dementia recruitment often fails when studies rely too heavily on tertiary centers alone. Community-connected networks can surface earlier-stage patients, maintain stronger local trust, and recruit through real-world referral flows instead of only specialty bottlenecks. When supported by good informed consent procedures, practical GCP compliance for coordinators, cleaner regulatory document management, and better patient recruitment companies and tech solutions, they often outperform more famous sites in pure enrollment practicality.
4. Academic Cognitive Neurology Centers
These sites stay near the top because biomarker-rich dementia studies, cognitively demanding endpoints, advanced imaging, and complex inclusion structures require deep scientific discipline. Strong centers create better alignment around clinical trial protocol management, sharper endpoint handling, cleaner data monitoring committee logic, and stronger clinical trial auditing and inspection readiness. Their weakness is speed. Academic rigor does not automatically mean rapid activation.
3. Movement Disorders Centers
Movement disorders sites earn this ranking because Parkinson’s and related studies depend heavily on rater experience, phenotype precision, visit timing discipline, medication-state awareness, and caregiver partnership. They often perform exceptionally when sponsors support them with better remote monitoring tools, clearer clinical trial documentation techniques, good research assistant documentation skills, and tight GCP compliance essentials for CRAs. Their biggest threat is portfolio competition across multiple Parkinson’s studies.
2. Integrated Neuroscience Hospital Research Units
These are powerful because they combine subspecialty neurology access, imaging, labs, inpatient relationships, outpatient continuity, and broader hospital infrastructure. For mixed neurology programs or complex multi-indication portfolios, integrated neuroscience units create resilience across startup and execution. They support stronger clinical trial project manager oversight, more stable clinical operations manager growth paths, better top regulatory compliance software use, and cleaner medical writing and document management control. Their challenge is complexity. Big systems can hide slow decisions.
1. Operationally Mature Academic–Community Hybrid Site Networks
The highest-ranked neurology site model in 2026 is the hybrid network that combines academic depth with community reach. This model wins because it balances scientific rigor, broader patient access, stronger caregiver logistics, indication-specific expertise, and more realistic enrollment execution. It performs best when supported by smart site qualification strategy, stronger clinical trial sponsor roles, tighter clinical trial staffing support, more targeted networking groups and forums, and better clinical research conferences and events. This model wins because neurology trials need both depth and reach, not one at the expense of the other.
3. The Capabilities That Actually Predict Neurology Trial Site Performance
The first predictor is diagnosis accuracy before formal screening. Neurology trials are unusually vulnerable to eligibility waste because many patients appear close to fit but collapse under protocol detail. A strong site does not merely have patients with the right disease label. It has investigators and coordinators who can narrow likely fits through chart review, medication history, disease-stage judgment, caregiver insight, and disciplined pre-screening logic. That improves patient recruitment efficiency, protects sample size planning, strengthens case report form accuracy, and reduces downstream protocol amendment pain.
The second predictor is endpoint discipline. Neurology outcomes are easily damaged by inconsistent cognitive testing, rater drift, timing errors, poor diary compliance, or visit-window slippage. A site can recruit well and still damage a study through noisy assessments. That is why strong teams examine rater training, backup raters, source documentation habits, caregiver instructions, and how the site handles blinding requirements, randomization integrity, cleaner clinical trial documentation, and active handling of clinical trial audits.
The third predictor is caregiver-aware operations. Many neurology studies do not recruit or retain a patient alone. They recruit and retain a family system. Sites that ignore transportation burden, caregiver work schedules, cognitive fatigue, visit length, and communication complexity often underperform despite strong medical teams. Better sites translate this into practical workflows through stronger informed consent best practices, clearer stakeholder communication, stronger patient safety oversight, and better clinical trial supply and support coordination.
The fourth predictor is whether the site can survive amendment shock. Neurology protocols often evolve as eligibility logic tightens, assessments shift, biomarker requirements change, or safety monitoring expands. A great neurology site is not just good before the first patient in. It is good after retraining, documentation changes, new operational burdens, and changing investigator expectations. That resilience is visible through stronger training requirements under GCP, good clinical research coordinator role discipline, sharper clinical research associate oversight, and more reliable quality assurance career-style thinking.
What is your biggest neurology clinical trial site-selection blocker right now?
Choose one. Your answer points to the fastest fix.
4. How to Match Neurology Site Types to the Right Indication
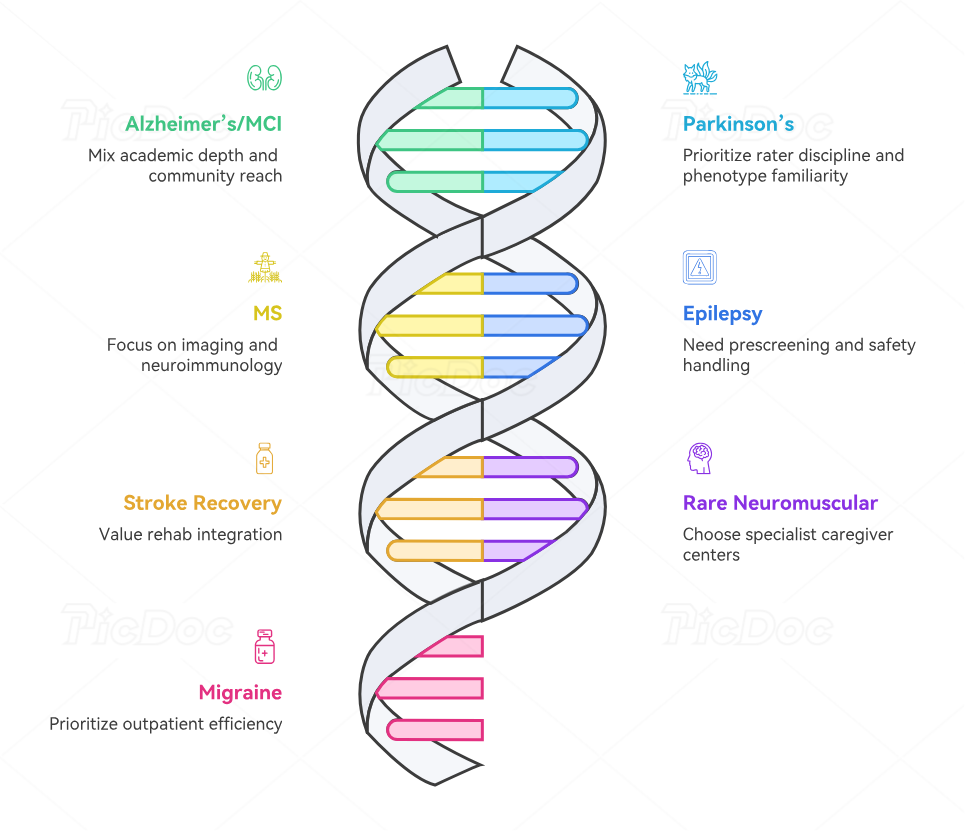
For Alzheimer’s disease and mild cognitive impairment, the winning site mix usually includes academic cognitive neurology depth plus community memory-clinic reach. You need sites that can handle caregiver-heavy screening, cognition-driven endpoints, biomarker logistics, and sensitive informed consent procedures, while still supporting broad enough funnels through better volunteer registries, smarter job portals and staffing pipelines, and practical clinical trial site and SMO recruiting ecosystems.
For Parkinson’s disease and related movement disorders, prioritize sites with proven rater discipline and deep phenotype familiarity. These sites need strong visit timing control, careful medication-state handling, and caregiver-sensitive follow-up. The best sponsor support here comes from tighter monitoring strategy, better site management, clearer clinical trial amendments handling, and smart use of clinical research networking groups and LinkedIn groups for clinical research professionals when building referral relationships and talent pipelines.
For MS studies, prioritize sites with imaging consistency, neuroimmunology experience, infusion capability when relevant, and long-term patient relationships. These studies benefit from strong clinical trial project management, more disciplined vendor oversight, tighter data management platform alignment, and more reliable remote monitoring platform support because imaging and longitudinal data cleanliness carry real weight.
For epilepsy, look for centers that can prescreen well, handle complex drug histories, and escalate safety cleanly. For stroke recovery, value rehabilitation integration and long-range retention operations more than acute-care prestige alone. For rare neuromuscular disease, choose specialist centers with caregiver infrastructure and realistic access to small populations rather than broad but shallow neurology sites. For migraine, prioritize outpatient efficiency, digital diary coaching, and communication precision. Across all of these, site selection improves when teams also strengthen continuing education use, read stronger clinical research journals and publications, and use relevant certification providers to sharpen staff capability where protocols are unusually demanding.
5. How Sponsors and CROs Should Build a Better Global Neurology Site Strategy
Start by abandoning the fantasy that “top global sites” can be chosen from reputation alone. Reputation helps, but site strategy should be built from capability clusters. Decide first whether your protocol needs volume, depth, biomarker rigor, rater precision, caregiver infrastructure, acute workflow strength, or long-duration retention excellence. Then map countries and site types accordingly. That approach creates stronger clinical operations leadership, sharper clinical trial manager roadmaps, more disciplined regulatory affairs support, and stronger clinical compliance officer thinking.
Second, qualify sites against failure scenarios, not only optimistic metrics. Ask how the site behaves when a key rater leaves, a caregiver misses a visit, an MRI slot is lost, a digital tool fails, or an amendment adds a new safety burden. The answers reveal far more than headline enrollment promises. This is where stronger clinical quality auditor instincts, better handling of protocol deviations, more practical research assistant data collection habits, and stronger medical monitor review thinking become strategically important.
Third, do not build a global neurology footprint that is all depth and no reach, or all reach and no depth. The best-performing portfolios mix flagship academic sites, practical community-connected feeders, and a few specialist depth centers for scientifically demanding cohorts. That balance improves resource allocation, reduces overdependence on one country or one star PI, creates better fallback options when feasibility fails, and supports stronger clinical research staffing models across the life of the study.
Finally, manage neurology sites like long-term partners, not interchangeable vendors. The best global sites want better forecasting, cleaner communication, realistic visit burden, sharper training, quicker escalation decisions, and less contradiction across sponsor, CRO, and central vendors. Sponsors and CROs that deliver that become easier to prioritize, easier to trust, and easier to enroll for. In neurology, trust is not soft. It is operational fuel.
6. FAQs
-
A high-value neurology site combines the right patient pool, indication-specific expertise, reliable raters, strong caregiver operations, consistent documentation, and realistic enrollment execution. The strongest sites also perform well under GCP guidelines, hold up during audit preparation, manage regulatory documents cleanly, and support clear patient recruitment workflows.
-
Academic centers are extremely valuable for biomarker-heavy, cognitively demanding, rare, or scientifically complex neurology trials. They are less universally dominant than many sponsors assume. Community-connected networks, outpatient specialty clinics, and hybrid systems often outperform them in pure enrollment practicality, caregiver logistics, or retention. The right answer depends on your protocol requirements, endpoint structure, study budget and resources, and risk management plan.
-
Rare neuromuscular diseases, complex dementia biomarker studies, advanced Parkinson’s trials, and some epilepsy programs usually need the most specialized sites. These studies depend on specialist diagnosis accuracy, advanced assessments, and tighter safety oversight. Teams should support them with stronger medical oversight, better drug safety processes, cleaner clinical trial monitoring, and more disciplined principal investigator responsibilities.
-
The biggest mistake is confusing general neurology prestige with protocol-specific fit. A site can have a brilliant PI and still underperform because its referral pathways, caregiver support, rater standardization, visit structure, or amendment resilience do not match the study. Better decisions come from stronger site qualification visits, sharper investigator site management, more realistic feasibility thinking through recruitment tools, and disciplined clinical trial sponsor oversight.
-
A strong portfolio usually includes a few flagship academic centers for scientific depth, several operationally mature community-connected sites for practical recruitment, and select specialist centers for rare or technically demanding cohorts. That balance protects against overreliance on a narrow site type and improves resilience across startup, enrollment, amendments, and retention. It also works better with broader clinical operations planning, cleaner vendor management, smarter resource allocation, and stronger quality oversight.
-
Ask about true pre-screening logic, caregiver dependence, backup raters, competing trial burden, amendment handling speed, digital compliance support, visit-window protection, and how the site rescues retention after missed visits. These answers often predict outcomes better than headline enrollment estimates. They also reveal whether the site is strong in GCP execution, documentation discipline, audit readiness, and patient safety oversight.